AOD 9604 is dosed at 300-500 mcg per day via subcutaneous injection on an empty stomach. This modified fragment of human growth hormone (amino acids 176-191) was developed for fat loss by Metabolic Pharmaceuticals in Australia. Six clinical trials involving approximately 900 subjects showed a favorable safety profile with side effects indistinguishable from placebo. The pivotal Phase 2b trial, however, failed to demonstrate statistically significant weight loss (Stier et al., Journal of Endocrinology and Metabolism).
| Quick Reference | Details |
|---|---|
| Full name | Advanced Obesity Drug 9604 |
| Structure | Modified HGH fragment (amino acids 176-191), tyrosine at N-terminal |
| Mechanism | Stimulates lipolysis via beta-3 adrenergic receptors, inhibits lipogenesis |
| Research dosage | 300-500 mcg/day subcutaneous |
| Serum half-life | Approximately 4 minutes (sustained enzyme activation) |
| Phase 2b result | Failed (2007): no significant weight loss vs placebo in 536 patients |
| Safety data | ~900 subjects across 6 RCTs, no serious adverse events |
| FDA status | Excluded from 503A compounding (2024) |
| WADA status | Banned in competition |
AOD 9604 mimics the lipolytic portion of growth hormone without affecting blood sugar or IGF-1 levels. The theoretical advantage over full HGH: fat loss without diabetogenic side effects. The practical problem: the Phase 2b clinical trial did not confirm efficacy in humans at therapeutic doses. For research on real-world results, see our AOD 9604 before and after guide.
This is educational content about a research compound. Consult a healthcare provider before using any peptide.
Get your custom peptide protocol:
- Tailored to your body and goals
- Precise dosing and cycle length
- Safe stacking combinations
- Backed by peer-reviewed studies
- Ready in under 2 minutes
AOD 9604 Dosage Chart
Research protocols follow body weight tiers. These doses come from community protocols and original clinical trial parameters, not from FDA-approved prescribing guidelines (none exist).
| Body Weight | Starting Dose (Weeks 1-2) | Standard Dose (Weeks 3-12) | Advanced Dose | Injection Volume (2,500 mcg/mL) |
|---|---|---|---|---|
| Under 150 lbs (68 kg) | 250 mcg | 300 mcg | 400 mcg | 0.10-0.16 mL |
| 150-200 lbs (68-91 kg) | 300 mcg | 400 mcg | 500 mcg | 0.12-0.20 mL |
| Over 200 lbs (91+ kg) | 300 mcg | 500 mcg | 500 mcg | 0.12-0.20 mL |
Timing. AOD 9604 is administered on an empty stomach, typically first thing in the morning. Insulin blunts AOD 9604's lipolytic activity because the peptide's mechanism depends on a low-insulin metabolic state. Wait at least 30 minutes before eating.
Split-dose protocol. Some protocols divide the daily dose: 250 mcg in the morning (fasted) and 250 mcg before bed (at least 3 hours after last meal). This approach maintains more consistent beta-3 receptor activation throughout the day.
Cycle length. Research protocols run 12 weeks (matching the Phase 2b trial duration). Some extend to 16 weeks. A 4-6 week off period between cycles is commonly recommended to prevent receptor desensitization.
Use our unit converter for dose calculations and see the peptide dosage chart for comparison with other peptides.
How to Reconstitute AOD 9604
AOD 9604 ships as a lyophilized (freeze-dried) powder. Standard reconstitution uses bacteriostatic water.
Standard preparation: 5 mg vial + 2 mL bacteriostatic water = 2,500 mcg/mL
| Desired Dose | Volume to Draw | Syringe Units (U-100) |
|---|---|---|
| 250 mcg | 0.10 mL | 10 units |
| 300 mcg | 0.12 mL | 12 units |
| 400 mcg | 0.16 mL | 16 units |
| 500 mcg | 0.20 mL | 20 units |
Alternative concentration: 5 mg vial + 1 mL bacteriostatic water = 5,000 mcg/mL (smaller injection volume, but harder to measure precise doses on standard syringes).
Steps: 1. Wipe both the vial stopper and bacteriostatic water vial with alcohol swabs. 2. Draw 2 mL of bacteriostatic water into a syringe. 3. Insert the needle into the AOD 9604 vial and let the water run down the inside wall. 4. Gently swirl the vial. Never shake peptides. 5. Wait until the powder dissolves completely (1-3 minutes).
Storage: Refrigerate at 2-8C. Use within 28-45 days after reconstitution. Do not freeze reconstituted peptides.
For detailed instructions, use our reconstitution calculator and see our guide on bacteriostatic water vs sterile water. For injection technique, see how to inject peptides.
How AOD 9604 Works
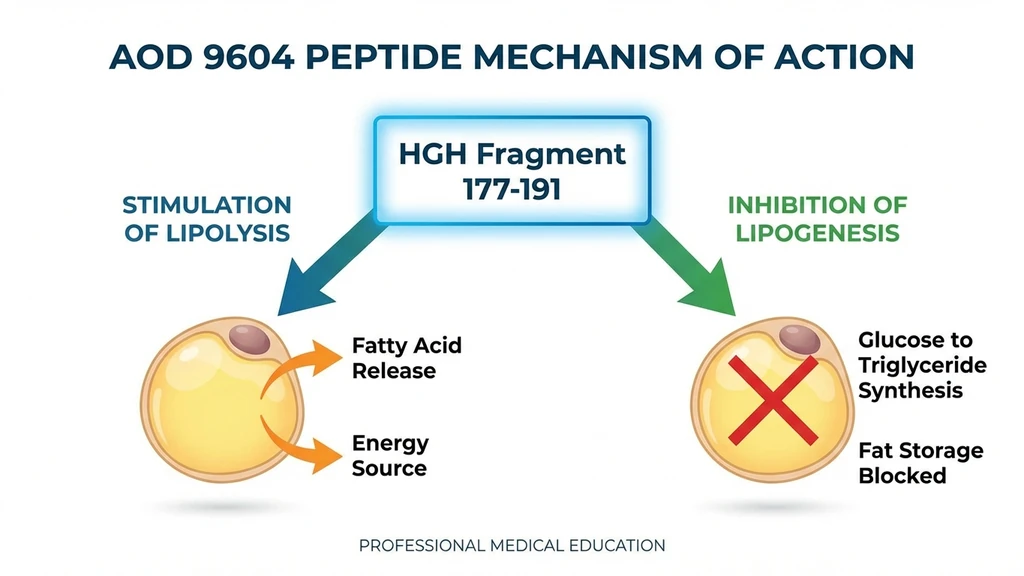
AOD 9604 is a 16-amino acid peptide derived from the C-terminal fragment of human growth hormone (residues 176-191). The key structural modification: a tyrosine residue replaces phenylalanine at the N-terminal, which preserves fat-metabolizing activity while eliminating growth-promoting effects.
Beta-3 adrenergic receptor activation. AOD 9604 upregulates beta-3 adrenergic receptor (beta-3 AR) expression on fat cells, triggering a cAMP signaling cascade that activates hormone-sensitive lipase. This enzyme breaks stored triglycerides into free fatty acids for energy use. Knockout mouse studies confirmed this pathway: beta-3 AR knockout mice were completely unresponsive to AOD 9604 (Heffernan et al., 2001, Endocrinology, PMID: 11713213).
Lipogenesis inhibition. AOD 9604 simultaneously reduces the rate at which excess calories convert to stored fat. In obese Zucker rats, 500 mcg/kg reduced weight gain by more than 50% (Ng et al., 2000, Hormone Research, PMID: 11146367).
Visceral fat selectivity. Animal data suggests particular efficacy on visceral adipose tissue, the metabolically dangerous fat surrounding organs.
Serum half-life. AOD 9604 has a very short serum half-life of approximately 4 minutes. This sounds limiting, but the peptide triggers sustained enzyme activation that continues after the peptide itself clears. This is why once-daily dosing produces measurable effects despite the short circulating half-life.
What it does NOT do. Unlike full HGH, AOD 9604 does not activate the IGF-1 pathway. No IGF-1 elevation means no blood sugar disruption, no cell proliferation risk, and no growth-related side effects. This was confirmed across all six clinical trials (Heffernan et al., 2001, International Journal of Obesity, PMID: 11673763).
Clinical Trial Evidence
No other dosage guide covers both the Phase 2a success and Phase 2b failure with this level of detail. Understanding both is essential for informed decision-making.
Phase 2a trial (positive results). Metabolic Pharmaceuticals enrolled 300 obese subjects across 5 Australian trial sites for 12 weeks. Six dose arms tested oral AOD 9604 at 1 mg, 5 mg, 10 mg, 20 mg, and 30 mg against placebo.
| Group | Dose (Oral) | Weight Loss | vs. Placebo |
|---|---|---|---|
| Placebo | N/A | -0.8 kg | N/A |
| Low dose | 1 mg | -2.8 kg | Significant |
| Higher doses | 5-30 mg | Variable | Dose-response inconsistent |
The 1 mg result looked promising. Metabolic Pharmaceuticals' stock surged on the announcement.
Phase 2b / OPTIONS study (failed). The larger confirmatory trial enrolled 536 obese patients for 24 weeks with oral dosing at 1 mg, 5 mg, and 10 mg.
| Group | Dose (Oral) | Weight Loss | vs. Placebo |
|---|---|---|---|
| Placebo | N/A | -2.0 kg | N/A |
| Low dose | 1 mg | -2.1 kg | Not significant |
| Medium dose | 5 mg | -2.3 kg | Not significant |
| High dose | 10 mg | -2.5 kg | Not significant |
The trial failed its primary endpoint. None of the AOD 9604 groups showed statistically significant weight loss compared to placebo. Development was terminated in 2007.
The oral bioavailability question. All human trials used oral administration. Peptide oral bioavailability is typically below 1%. AOD 9604 is unusual: animal data shows approximately 40% oral bioavailability (Ng et al., PMID: 11146367). Subcutaneous injection achieves near-100% bioavailability. Injectable AOD 9604 has never been tested in a controlled human trial. Community use is primarily subcutaneous, a different route than what was clinically tested. This is the critical gap in the evidence.
Safety across all 6 trials (~900 subjects). No treatment-related serious adverse events. No clinically significant adverse events. No anti-AOD9604 antibodies detected (immunogenicity negative across all subjects tested). No IGF-1 changes. Adverse event profile indistinguishable from placebo (Stier et al., Journal of Endocrinology and Metabolism).
AOD 9604 Side Effects
In six randomized, double-blind, placebo-controlled trials involving approximately 900 adults, AOD 9604's adverse event profile was indistinguishable from placebo.
| Side Effect | Frequency | Severity | Duration | Notes |
|---|---|---|---|---|
| Injection site redness | Common | Mild | 1-2 hours | Standard for subcutaneous peptides |
| Injection site swelling | Common | Mild | 1-2 hours | Rotate injection sites |
| Headache | Occasional | Mild-moderate | First 1-2 weeks | Typically resolves with continued use |
| Nausea | Rare | Mild | Transient | More common at higher doses |
| Flu-like symptoms | Rare | Mild | First 2-3 days | Immune system adjustment |
| Fatigue | Rare | Mild | Transient | Reported anecdotally, not confirmed in trials |
What AOD 9604 does NOT cause (confirmed in clinical data):
- No blood sugar changes. Unlike full HGH, AOD 9604 does not affect glucose metabolism.
- No IGF-1 elevation. "AOD9604 had no effect on serum IGF-1 levels" across all trials.
- No antibody formation. Immunogenicity testing was negative in all subjects tested across 6 trials.
- No water retention. Unlike full HGH, which causes significant edema in many users.
- No insulin resistance. Full HGH is diabetogenic; AOD 9604 is not.
- No cortisol or HPA axis changes.
Contraindications and cautions:
- Pregnancy and breastfeeding (no safety data exists)
- Active cancer (theoretical concern with any growth-hormone-derived compound)
- Under 18 years old (no pediatric data)
- Concomitant full HGH use (redundant pathways, no additive benefit expected)
- Athletes in WADA-tested competition (prohibited substance)
The safety profile is the strongest argument for AOD 9604. The weakness is on the efficacy side, not the safety side. For comparison with other fat-loss approaches, see our tesamorelin dosage guide and sermorelin guide.
AOD 9604 vs Semaglutide for Weight Loss
This is the most common comparison question. The honest answer: semaglutide has vastly superior evidence.
| Feature | AOD 9604 | Semaglutide (Wegovy) |
|---|---|---|
| Mechanism | Beta-3 AR lipolysis (fat cells) | GLP-1 receptor agonist (brain + gut) |
| FDA approved | No | Yes (June 2021) |
| Largest trial | 536 patients (failed primary endpoint) | 1,961 patients (STEP 1, succeeded) |
| Weight loss | Not significant vs placebo | 14.9% body weight at 68 weeks |
| Administration | Daily subcutaneous injection | Weekly subcutaneous injection |
| Side effects | Indistinguishable from placebo | 44% nausea, 31% diarrhea |
| Appetite suppression | None | Strong |
| Monthly cost | $40-80 (research grade) | $199-499 (compounded) |
| Muscle preservation | Neutral | Some lean mass loss |
| Cardiovascular benefit | Not studied | 20% MACE reduction (SELECT trial) |
AOD 9604's advantages are limited to three areas: lower side effects, lower cost, and no appetite suppression (some users prefer maintaining normal appetite). For patients seeking proven pharmaceutical fat loss, semaglutide and tirzepatide are backed by large randomized controlled trials showing 15-22% weight loss.
See our semaglutide dosage chart and tirzepatide dosage chart for FDA-approved weight loss medication dosing. Compare costs with our cost calculator.
AOD 9604 for Joint Health and Cartilage Repair
An overlooked secondary application supported by animal research and Australian regulatory approval.
Kwon and Park (2015) tested intra-articular AOD 9604 combined with hyaluronic acid in a rabbit osteoarthritis model. The combination enhanced cartilage regeneration significantly beyond either treatment alone. AOD 9604 stimulated chondrocyte proliferation and increased type II collagen production, the primary structural protein in articular cartilage (Annals of Clinical and Laboratory Science, PMID: 26275694).
The Australian Therapeutic Goods Administration (TGA) approved AOD 9604 for cartilage repair formulations, recognizing its chondroprotective properties separately from its failed fat-loss development.
This application uses intra-articular injection (directly into the joint), not the subcutaneous route used for fat loss. Dosing protocols for cartilage repair differ from the fat-loss protocols described above. No controlled human trial has confirmed cartilage repair efficacy.
For more on peptides used for joint recovery, see our best peptides for joint pain guide.
Stacking AOD 9604 With Other Peptides
AOD 9604 can be combined with peptides that use different mechanisms. Verify combinations with our interaction checker and plan protocols with our stack calculator.
AOD 9604 + CJC-1295/Ipamorelin. Fat loss synergy through complementary pathways. AOD 9604 targets fat cells directly (beta-3 AR). CJC-1295 and Ipamorelin stimulate growth hormone release, which promotes fat metabolism through a different cascade. Timing: AOD 9604 in the morning (fasted), CJC/Ipamorelin before bed (to amplify natural nighttime GH pulse).
AOD 9604 + BPC-157. Fat loss combined with tissue repair. BPC-157 works through nitric oxide and growth factor pathways unrelated to beta-3 adrenergic receptors. No known interaction. Can be administered in the same syringe or separately.
AOD 9604 + Semaglutide. Complementary mechanisms: AOD 9604 targets fat cells directly while semaglutide suppresses appetite through GLP-1 receptors. No published interaction data suggests contraindication. This is a "belt and suspenders" approach.
AOD 9604 + HGH Fragment 176-191. Do not stack. AOD 9604 IS a modified version of HGH Frag 176-191. Both target the same beta-3 AR pathway. Combining them provides no additive benefit and wastes product.
FDA and Legal Status (2026)
AOD 9604 occupies a complicated and evolving regulatory space.
FDA 503A exclusion (2024). The FDA added AOD 9604 to the list of substances that cannot be compounded under Section 503A. Licensed compounding pharmacies in the United States cannot legally prepare AOD 9604 for individual patients. The FDA cited insufficient safety and efficacy data for therapeutic use.
PCAC advocacy (December 2024). The Pharmacy Compounding Advisory Committee heard testimony from advocates including Dr. Edwin Lee, who presented safety data from the 900-patient clinical dataset arguing that AOD 9604 should remain available for compounding. The committee's recommendation was advisory only.
GRAS status (2007). The FDA granted AOD 9604 Generally Recognized As Safe status as a food ingredient. This is NOT therapeutic approval. GRAS means it is safe to consume in food products at specified levels. Many sources incorrectly conflate GRAS food status with drug safety approval.
WADA prohibited. The World Anti-Doping Agency bans AOD 9604 under "Peptide Hormones, Growth Factors, Related Substances and Mimetics." Athletes face sanctions for positive tests.
Research chemical availability. AOD 9604 remains available from research suppliers labeled "for research use only." This gray market is where most users acquire the peptide. Research chemicals are not regulated as drugs and are technically not legal for human self-administration.
Australian TGA. In Australia, AOD 9604 was approved for cartilage repair formulations by the Therapeutic Goods Administration.
For the broader regulatory landscape, see our peptide legality guide and FDA peptide crackdown analysis.
AOD 9604 vs Other Fat-Loss Peptides
| Peptide | Mechanism | Efficacy Evidence | Side Effects | Monthly Cost |
|---|---|---|---|---|
| AOD 9604 | HGH fragment (beta-3 AR lipolysis) | Phase 2b failed | Minimal | $40-80 |
| Semaglutide | GLP-1 agonist | FDA-approved (14.9% loss) | GI (44% nausea) | $199-499 |
| Tirzepatide | GLP-1/GIP dual agonist | FDA-approved (22.5% loss) | GI (31% nausea) | $299-499 |
| Tesamorelin | GHRH analog (GH release) | FDA-approved (lipodystrophy) | Moderate | $200-400 |
| CJC-1295/Ipamorelin | GH secretagogue | Limited human data | Moderate | $100-200 |
| MOTS-c | Mitochondrial peptide | Preclinical only | Unknown | $80-150 |
AOD 9604 is the lowest-cost option with the weakest clinical evidence. Semaglutide and tirzepatide have FDA approval, large RCTs, and proven 15-22% weight loss. For proven pharmaceutical fat loss, GLP-1 drugs are the clear choice. AOD 9604 remains in the research conversation because of its favorable safety profile and low cost.
See our best peptides for weight loss guide for a comprehensive comparison.
Frequently Asked Questions
Does AOD 9604 actually work for fat loss?
The Phase 2b clinical trial (536 patients, oral dosing, 24 weeks) showed no statistically significant weight loss compared to placebo. An earlier Phase 2a trial (300 patients) showed modest results at 1 mg oral. Animal studies show positive lipolysis effects. Injectable AOD 9604 has never been tested in a controlled human trial, so subcutaneous efficacy remains unproven.
What is the correct AOD 9604 dosage?
Research protocols use 300-500 mcg per day via subcutaneous injection on an empty stomach. Weight-based approach: 300 mcg for under 150 lbs, 400 mcg for 150-200 lbs, 500 mcg for over 200 lbs. A split-dose protocol (250 mcg morning, 250 mcg before bed) is an alternative. No FDA-approved dosing exists.
When should I inject AOD 9604?
First thing in the morning, at least 30 minutes before eating. Insulin blunts AOD 9604's lipolytic activity by suppressing the beta-3 AR cascade, so a fasted state is essential. Some protocols add a second dose before bed, also at least 3 hours after the last meal.
Is AOD 9604 the same as HGH Fragment 176-191?
Nearly identical but not the same. AOD 9604 has a tyrosine residue at the N-terminal instead of phenylalanine. Both derive from the C-terminal of human growth hormone and target beta-3 adrenergic receptors. They should not be stacked together because they act on the same pathway.
Is AOD 9604 safe?
Six clinical trials involving approximately 900 subjects showed an adverse event profile indistinguishable from placebo. No serious adverse events were attributed to AOD 9604. No anti-AOD9604 antibodies were detected (immunogenicity negative). It does not affect IGF-1, blood sugar, or insulin sensitivity (Stier et al., Journal of Endocrinology and Metabolism).
Is AOD 9604 legal in the United States?
The FDA excluded AOD 9604 from 503A compounding in 2024. Licensed pharmacies cannot compound it for patients. It is banned by WADA. It remains available from research chemical suppliers for laboratory use only. The FDA separately granted it GRAS status as a food ingredient, which is not therapeutic approval.
How long does an AOD 9604 cycle last?
Standard research protocols run 12 weeks, matching the Phase 2b clinical trial duration. Some extend to 16 weeks. A 4-6 week off period between cycles is commonly recommended to prevent receptor desensitization, though no published data establishes the optimal cycling schedule.
AOD 9604 vs semaglutide: which is better for weight loss?
Semaglutide has vastly superior evidence: FDA-approved, with 14.9% body weight loss over 68 weeks in the STEP 1 trial (1,961 patients). AOD 9604 failed its Phase 2b trial. Semaglutide causes more side effects (44% nausea, 31% diarrhea) but produces dramatically greater weight loss. AOD 9604's advantages are lower cost ($40-80 vs $199-499/month) and minimal side effects.
The Bottom Line
AOD 9604 is a research peptide with a strong theoretical mechanism, a 900-patient safety record, and a failed Phase 2b clinical trial. The FDA excluded it from compounding, and WADA banned it from competition.
The safety profile is genuinely favorable: no significant adverse effects, no blood sugar disruption, no IGF-1 elevation, no antibody formation. For a peptide that has not demonstrated clinical efficacy, it at least does not cause harm.
The unresolved question is oral vs. injectable bioavailability. All human trials used oral dosing. Subcutaneous injection achieves near-100% bioavailability versus approximately 40% oral. Whether this difference would change clinical outcomes remains untested.
For proven fat loss, semaglutide and tirzepatide have FDA approval backed by large clinical trials showing 15-22% weight loss. Use our cost calculator for treatment planning and our reconstitution calculator for preparation guidance.
This is educational content about a research compound. Consult a healthcare provider before using any peptide.
Related Articles
BPC-157 Dosage Per Body Weight (Chart)
BPC-157 dosage by body weight: 1.6-10 mcg/kg. Full chart for 120-280 lbs, injection vs oral doses, and why most use flat 250-500 mcg.
How Much Sermorelin Per Day: Complete Dosage Guide
Sermorelin dosage: 200-500 mcg/day subcutaneous before bedtime. Starting dose, titration, gender-specific protocols, and IGF-1 monitoring.
BPC-157 TB-500 Dosage: Combined Protocol
BPC-157 TB-500 dosage: 250-500 mcg BPC-157 + 2-2.5 mg TB-500 daily. Weight-adjusted charts, injury-type matrix, blend vial math, and cycle timing.
Semaglutide Mixing Chart: Every Vial Size (2026)
Semaglutide mixing chart for 3mg, 5mg, 10mg, 15mg vials. BAC water ratios, unit conversion, step-by-step reconstitution, titration schedule, and storage guide.