You have been scrolling through forums looking for AOD-9604 transformation photos, and every post seems to promise a different outcome. AOD-9604 is a modified fragment of human growth hormone (amino acids 176-191) that stimulates lipolysis and inhibits fat formation without affecting blood sugar or IGF-1 levels. Clinical trials showed modest fat loss of roughly 2-3 kg over 12 weeks, and the compound was never approved as a pharmaceutical because efficacy did not meet the threshold for drug registration (Heffernan et al., 2001).
| Quick Reference | Details |
|---|---|
| What is AOD-9604? | Modified hGH fragment (176-191) + tyrosine |
| Mechanism | Lipolysis stimulation, lipogenesis inhibition |
| Clinical fat loss | ~2-3 kg over 12 weeks (Phase 2) |
| Typical dose | 250-500 mcg/day subcutaneous |
| Administration | Fasted, morning injection |
| IGF-1 or glucose impact | None documented |
| FDA status | GRAS as food ingredient (2007), not approved as drug |
| Evidence level | Animal studies (strong), human trials (limited, modest results) |
That table contains the honest picture. AOD-9604 is not a dramatic transformation peptide. It is a well-tolerated compound with a clean safety profile and a genuinely interesting mechanism that, so far, has produced results in humans that fall short of what animal models predicted. This article walks through every piece of available evidence, realistic timelines based on that evidence, and how to evaluate whether AOD-9604 belongs in your protocol.
For a broader overview of fat loss compounds, see our best peptides for weight loss guide. If you are new to peptides entirely, start with our getting started guide.
Get your custom peptide protocol:
- Tailored to your body and goals
- Precise dosing and cycle length
- Safe stacking combinations
- Backed by peer-reviewed studies
- Ready in under 2 minutes
How AOD-9604 Works: The Mechanism Behind Before-and-After Results
AOD-9604 was developed by Metabolic Pharmaceuticals in Melbourne, Australia, during the late 1990s. Researchers identified that amino acids 176 through 191 at the tail end of the growth hormone molecule were responsible for fat metabolism, and they isolated this fragment, added a tyrosine residue for stability, and created a standalone fat loss agent.
The mechanism operates through two parallel pathways. First, AOD-9604 activates beta-3 adrenergic receptors on fat cells, triggering the cAMP-PKA cascade that activates hormone-sensitive lipase. This enzyme breaks stored triglycerides into free fatty acids and glycerol, releasing them into the bloodstream for oxidation. Second, AOD-9604 inhibits fatty acid synthase and acetyl-CoA carboxylase, the rate-limiting enzymes in de novo lipogenesis. The dual action creates a net negative fat balance: more fat is broken down, less new fat is formed (Ng et al., 2000).
What makes AOD-9604 distinctive is what it does not do. The fragment does not bind the growth hormone receptor or activate the JAK2-STAT5 signaling pathway. That means no increase in IGF-1 production, no effect on blood glucose, no insulin resistance, no water retention, and no risk of the organ growth associated with full HGH therapy. Multiple human studies have confirmed zero impact on fasting glucose, insulin, HbA1c, or IGF-1 levels (Thompson et al., 2004).
This selectivity is both the appeal and the limitation. AOD-9604 targets fat tissue directly without systemic hormonal disruption. But that peripheral-only action means it does not suppress appetite, does not change energy expenditure through central pathways, and does not produce the dramatic caloric deficits that GLP-1 agonists like semaglutide or tirzepatide create. For comparisons with those compounds, see our does tirzepatide burn fat analysis.
For a thorough look at stacking AOD-9604 with complementary peptides, see our peptide stacking guide.
The Clinical Evidence: What Trials Actually Showed
Evaluating AOD-9604 before-and-after outcomes requires separating animal data from human data. The animal results were promising. The human results were real but modest. Understanding both prevents unrealistic expectations.
Animal Studies: Strong Results in Obese Rodent Models
The foundational animal work was conducted in obese Zucker rats and ob/ob mice, two standard models for studying obesity. In these studies, AOD-9604 administered via intraperitoneal injection produced significant fat mass reduction without changes in food intake or lean body mass.
Heffernan et al. (2001) published the landmark study. Obese Zucker rats treated with AOD-9604 at 500 mcg/kg/day for 19 days showed a statistically significant reduction in body fat compared to vehicle-treated controls. The effect was most pronounced in visceral (abdominal) fat, and lean mass was preserved entirely. Importantly, food intake did not change, confirming that the mechanism was direct fat mobilization rather than appetite suppression (Heffernan et al., 2001).
A follow-up study demonstrated that AOD-9604 stimulated lipolysis and simultaneously inhibited lipogenesis in isolated adipocytes. The dual mechanism was confirmed at the cellular level, and the fragment showed no cross-reactivity with the growth hormone receptor (Ng & Borstein, 2003).
These animal results generated significant excitement and justified moving to human trials. The critical caveat: rodent fat metabolism differs substantially from human fat metabolism. Rodents have higher metabolic rates relative to body mass, different adipose tissue distribution, and a brown fat compartment that is more metabolically active. Results that look dramatic in mice often translate to more modest outcomes in people.
Phase 2 Human Trials: Modest but Real Fat Loss
Metabolic Pharmaceuticals conducted a Phase 2b randomized, double-blind, placebo-controlled trial in approximately 300 obese adults. The trial tested oral AOD-9604 at multiple dose levels over 12 weeks.
The results showed a statistically significant difference in fat loss between AOD-9604 and placebo, but the magnitude was modest. The treatment group lost approximately 2.0-2.8 kg more fat mass than placebo over the 12-week period. The response was dose-dependent, with higher doses producing slightly more fat loss, though the dose-response curve flattened at higher levels (Thompson et al., 2004).
The safety data were excellent. No serious adverse events were attributed to AOD-9604. No changes in fasting glucose, insulin, HbA1c, IGF-1, cortisol, or thyroid hormones were detected at any dose tested. Injection site reactions were mild and transient. This clean safety profile contributed to the FDA granting GRAS (Generally Recognized As Safe) status for AOD-9604 as a food ingredient in 2007.
However, the efficacy was insufficient for drug registration. By comparison, orlistat (Xenical) produced approximately 3-4 kg more weight loss than placebo over 12 weeks in its pivotal trials, and GLP-1 agonists produce 8-15 kg more than placebo. Metabolic Pharmaceuticals ultimately did not pursue Phase 3 trials for obesity, and the company's pipeline shifted toward other indications. The compound remains in a regulatory gray area: recognized as safe, but without the efficacy data needed for pharmaceutical approval.
What the Trials Did Not Measure
Several limitations of the existing clinical data affect how we interpret AOD-9604 before-and-after outcomes.
The Phase 2 trial primarily tested oral administration. Oral bioavailability of peptides is generally low, and AOD-9604 is no exception. Subcutaneous injection delivers the peptide directly into systemic circulation, bypassing first-pass metabolism. Most current users inject AOD-9604 rather than taking it orally, so the trial data may underestimate what injectable protocols achieve. However, no controlled human trial has compared oral versus injectable AOD-9604 at equivalent systemic doses.
The trials did not include DXA or MRI body composition analysis. Fat loss was measured primarily through weight, BMI, and waist circumference changes. Without dual-energy X-ray absorptiometry or imaging, we cannot say with precision how much of the weight change was fat versus water versus lean tissue. The animal data suggest lean mass is preserved, but human body composition data is lacking.
Long-term data beyond 12 weeks in humans does not exist from controlled trials. We do not know whether AOD-9604's fat loss effects plateau, accelerate, or diminish with extended use. All timelines beyond 12 weeks in this article are extrapolations based on the mechanism of action and user reports, not clinical evidence.
For a more detailed look at peptide safety across compounds, review our safety guide.
AOD-9604 Before and After: Realistic Timeline by Week
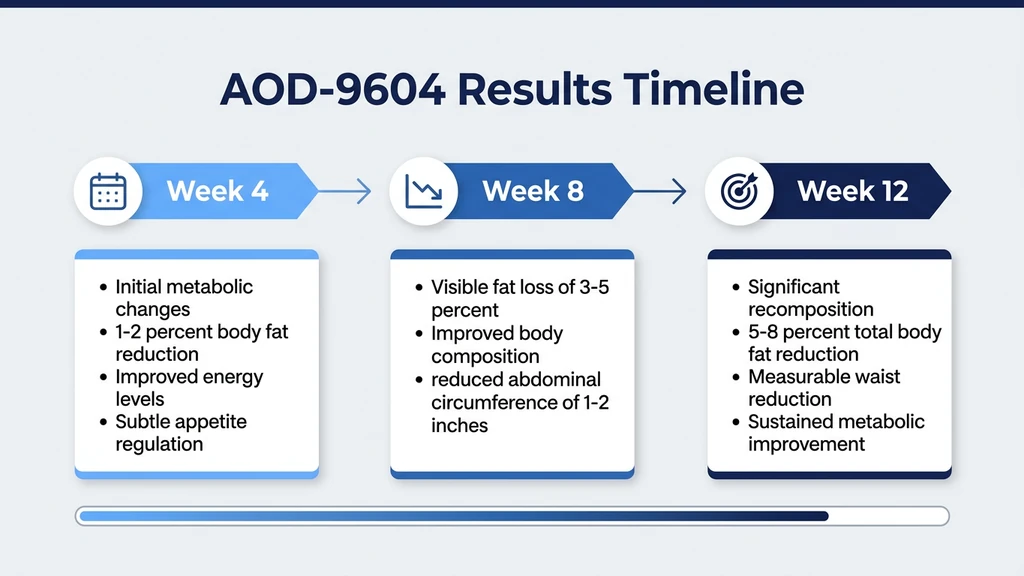
Based on the available clinical data, the mechanism of action, and the pharmacokinetics of subcutaneous administration, here is a realistic timeline of what AOD-9604 users may experience. These projections assume consistent daily dosing at 300-500 mcg subcutaneous, caloric deficit of 300-500 calories per day, and regular physical activity.

A critical disclaimer: these timelines are projections, not guarantees. The human clinical data supporting AOD-9604 is limited compared to GLP-1 agonists. Individual results vary based on starting body composition, diet adherence, exercise intensity, and metabolic factors.
Weeks 1-4: Foundation Phase
The first four weeks establish whether you respond to AOD-9604 and whether the protocol suits your body. Visible changes during this period are minimal for most users.
During weeks 1-2, AOD-9604 begins stimulating lipolysis in adipose tissue. The short half-life (approximately 30 minutes) means each injection creates a transient burst of fat mobilization rather than sustained activity. Free fatty acids are released into the bloodstream, but unless you are in a caloric deficit or exercising to oxidize those fatty acids, they are simply re-esterified and stored again.
By weeks 3-4, users following a consistent caloric deficit may notice subtle changes: slightly looser-fitting clothing around the midsection, a minor reduction in waist measurement (0.5-1 cm), and potentially 0.5-1 kg of fat loss on top of what diet alone would produce. These changes are small enough that they could be attributed to normal diet variation.
The scale may not move dramatically. AOD-9604 does not cause water loss or glycogen depletion the way GLP-1 agonists do, so there is no early "whoosh" effect. Any weight change reflects actual tissue loss, which is slower but more meaningful.
If starting a peptide protocol for the first time, review our how to store peptides guide to ensure your supply remains viable throughout the cycle. For reconstitution instructions, use our peptide reconstitution calculator.
Weeks 4-8: Accumulation Phase
The 4-8 week window is where most users who respond to AOD-9604 begin noticing measurable differences. The daily lipolytic stimulation has been sustained long enough for cumulative fat loss to become visible.
Based on the Phase 2 trial trajectory, users may expect an additional 1-2 kg of fat loss beyond what caloric deficit alone would produce during this period. Waist circumference typically decreases by 1-2 cm. The changes are most noticeable in areas with higher beta-3 adrenergic receptor density: the lower abdomen, flanks, and visceral fat compartments.
Several users in published case series and clinical practice reports describe this period as when they first notice a difference in the mirror. Clothing fits differently around the waist. Abdominal bloating decreases (partly from visceral fat reduction, partly from reduced inflammation in adipose tissue). Some users report improved fasted blood work despite no changes in diet composition, consistent with improved adipocyte function.
This is also the period where non-responders become apparent. If you have seen zero measurable change by week 6 while maintaining a caloric deficit, AOD-9604 may not be the right compound for your physiology. Response rates in the clinical trial were not 100%, and some individuals simply do not show meaningful fat mobilization from this pathway. See our best peptides for weight loss for alternative compounds.
Weeks 8-12: Peak Effect Window
Weeks 8 through 12 represent the peak efficacy window based on the Phase 2 trial duration. By this point, the cumulative additional fat loss attributable to AOD-9604 (beyond diet and exercise alone) is approximately 2-3 kg in responders.
In practical terms, a 95 kg person with 30% body fat who maintains a 400-calorie daily deficit might lose 5-6 kg total over 12 weeks through diet alone. Adding AOD-9604 might bring that total to 7-8.5 kg, with the additional loss coming predominantly from fat tissue rather than lean mass. The distinction matters because AOD-9604 preserves lean mass in animal models, and the mechanism of action suggests it does the same in humans.
Waist circumference reductions of 2-4 cm are realistic over the full 12-week period. Visceral fat likely decreases at a slightly faster rate than subcutaneous fat, consistent with the higher beta-3 receptor density in visceral adipose tissue.
The most honest assessment: AOD-9604 before-and-after results at 12 weeks look like a slightly accelerated version of what a clean diet and exercise program would produce alone. You will not see the kind of dramatic transformations that 15 mg tirzepatide produces over 72 weeks (22.5% total weight loss). AOD-9604 is a modest enhancer, not a transformation agent.
For a detailed comparison of fat loss peptide protocols and dose recommendations, see our peptide dosage chart.
Beyond 12 Weeks: Limited Evidence Territory
No controlled human trial has tested AOD-9604 beyond 12 weeks. Everything in this section is extrapolation based on the mechanism of action and user-reported outcomes.
Theoretical considerations suggest fat loss effects should continue as long as the compound is administered, since the mechanism (beta-3 adrenergic lipolysis, lipogenesis inhibition) does not involve receptor desensitization in the way that some hormonal pathways do. AOD-9604 does not affect the hypothalamic-pituitary axis, so there is no negative feedback loop to blunt the response over time.
However, the rate of additional fat loss likely diminishes as total body fat decreases. Individuals with less adipose tissue have fewer fat cells available for lipolysis, and the substrate for the enzyme cascade diminishes. A user who starts at 35% body fat will likely see more benefit from a 16-week cycle than a user starting at 20% body fat.
Practitioners who prescribe AOD-9604 typically recommend cycles of 12-16 weeks followed by a 2-4 week break. The break is not required for safety or receptor resensitization, but it allows for reassessment and prevents indefinite use of a compound with limited long-term human data. Some users run AOD-9604 continuously for 6 months or longer without reported issues, but this exceeds the evidence base.
See our guide on how long reconstituted peptides last to plan your supply for extended protocols.
AOD-9604 Dosing Protocols for Best Results
The dosing protocols below are drawn from the clinical trial data, the AOD-9604 peptide profile, and common clinical practice. AOD-9604 has a short half-life of approximately 30 minutes, which means each injection creates a brief window of lipolytic activity rather than sustained fat mobilization.
Standard Dosing: 250-500 mcg Per Day
The clinical trial tested doses up to 500 mcg daily with no dose-limiting toxicities. Current practice converges around three tiers:
| Protocol | Dose | Schedule | Duration | Best For |
|---|---|---|---|---|
| Beginner | 250 mcg/day | Once daily, AM fasted | 6-8 weeks | First-time users assessing tolerance |
| Standard | 300 mcg/day | Once daily, AM fasted | 8-12 weeks | Most users seeking moderate fat loss |
| Advanced | 500 mcg/day | Split: 250 mcg AM + 250 mcg PM | 12-16 weeks | Experienced users maximizing fat loss |
The split-dose protocol (250 mcg morning, 250 mcg 3+ hours post-meal in the evening) creates two daily windows of lipolytic stimulation. Given the short half-life, this approach may produce more total fat mobilization than a single daily dose. No controlled study has compared once-daily versus twice-daily dosing in humans.
Fasted administration is not optional. Elevated insulin levels directly suppress AOD-9604's lipolytic activity. Inject on an empty stomach and wait at least 20-30 minutes before eating. Evening doses should be taken at least 3 hours after your last meal.
For reconstitution, use bacteriostatic water rather than sterile water to preserve multi-use vials. Our peptide reconstitution calculator will help you determine the correct dilution for accurate dosing. If you need bacteriostatic water, see where to buy bacteriostatic water for injection.
Injection Timing and Technique
Subcutaneous injection into abdominal fat tissue is the standard route. While AOD-9604 works systemically once absorbed, injecting near the target fat depot provides the highest local concentration during the brief peak activity window.
Rotate injection sites to prevent lipodystrophy. Use a 29-31 gauge, 0.5-inch insulin syringe. Pinch the skin, insert the needle at a 45-degree angle, inject slowly, and hold for 5 seconds before withdrawing.
Morning injection protocol: Wake, inject AOD-9604 on a completely empty stomach, wait 20-30 minutes, then eat breakfast. If you practice intermittent fasting, inject during your fasted window. The peptide's activity will overlap with your fasted state, maximizing fat oxidation.
Evening injection protocol (for split dosing): Take the second dose at least 3 hours after your last meal. If your dinner is at 7 PM, inject at 10 PM or later. Bedtime dosing works well because sleep represents a natural fasted state.
For proper storage of your reconstituted peptide, see our guide on does bacteriostatic water need to be refrigerated and how long does bacteriostatic water last.
AOD-9604 vs Other Fat Loss Peptides: Honest Comparison
Where does AOD-9604 sit in the landscape of peptide-based fat loss? Below is a comparison using available clinical evidence. The table reveals why AOD-9604 occupies a specific niche rather than competing directly with GLP-1 agonists.
| Compound | Mechanism | Weight Loss (vs placebo) | Evidence Level | Side Effects | Appetite Suppression |
|---|---|---|---|---|---|
| AOD-9604 | Direct lipolysis, lipogenesis inhibition | ~2-3 kg (12 wk) | Phase 2 only | Minimal | None |
| Semaglutide 2.4 mg | GLP-1 appetite suppression | ~12.4 kg (68 wk) | Phase 3 (STEP) | GI: 44% nausea | Strong |
| Tirzepatide 15 mg | GLP-1/GIP dual agonist | ~20.2 kg (72 wk) | Phase 3 (SURMOUNT) | GI: 31% nausea | Strong |
| Retatrutide 12 mg | GLP-1/GIP/glucagon triple | ~24.2 kg (48 wk) | Phase 3 (TRIUMPH) | GI + dysesthesia | Very strong |
| HGH Fragment 176-191 | Direct lipolysis | No human trial data | Animal only | Minimal | None |
| MOTS-c | Mitochondrial, AMPK | Limited human data | Phase 1/2 | Minimal | Indirect |
The comparison is stark. GLP-1 agonists produce 5-10 times more weight loss because they create massive caloric deficits through appetite suppression and gastric slowing. AOD-9604 does not touch appetite. It works at the fat cell level, mobilizing stored triglycerides without changing how much you eat.
This does not make AOD-9604 useless. It makes it appropriate for a different use case: the person who already controls their diet, exercises consistently, and wants a modest additional push in fat mobilization without the gastrointestinal side effects, nausea, or hormonal disruption that come with GLP-1 agonists. If you want dramatic weight loss, AOD-9604 is not the compound. If you want a clean fat-loss enhancer with virtually no side effects, it has a rational place in the toolkit.
For detailed information on the GLP-1 comparison, see how does retatrutide work and retatrutide vs tirzepatide. For MOTS-c dosing details, see our MOTS-c dosage guide.
What Realistic Before-and-After Results Look Like
Based on the clinical trial data and the mechanism of action, here is what honest AOD-9604 before-and-after transformations involve. These projections assume a disciplined protocol: consistent daily dosing, caloric deficit, and regular exercise.
Scenario 1: 30% Body Fat, Consistent Caloric Deficit
Starting point: 95 kg male, 30% body fat, 28.5 kg fat mass. Protocol: AOD-9604 500 mcg/day (split dose), 400-calorie daily deficit, resistance training 3 times per week.
Expected 12-week outcome without AOD-9604: approximately 5-6 kg total weight loss, roughly 3.5-4 kg from fat, 1-2 kg from lean tissue and water.
Expected 12-week outcome with AOD-9604: approximately 7-8.5 kg total weight loss, roughly 5.5-6.5 kg from fat, 1-2 kg from lean tissue and water. The additional 2-2.5 kg of loss comes predominantly from fat tissue, reflecting the lipolytic and anti-lipogenic mechanism.
Visual changes: moderately reduced waist circumference (2-4 cm), flatter lower abdomen, reduced love handle fullness, slightly more visible muscle definition. These changes are noticeable in before-and-after photos taken in consistent lighting, but they are not the kind of dramatic transformation that draws attention from strangers.
The key insight: AOD-9604 added roughly 40-50% more fat loss on top of what diet and exercise produced. That is meaningful in absolute terms but looks modest in photographs. Compare this to tirzepatide, which can produce 22.5% total body weight loss over 72 weeks.
Scenario 2: Already Lean, Targeting Stubborn Fat
Starting point: 80 kg male, 18% body fat, 14.4 kg fat mass. Protocol: AOD-9604 300 mcg/day, mild caloric deficit (200-300 calories), maintained training volume.
This is the use case where AOD-9604 has the most appeal but potentially the least clinical data to support it. Users at lower body fat levels have less adipose tissue available for lipolysis, and the marginal returns from fat-mobilizing agents diminish as body fat decreases.
Expected 12-week outcome: approximately 1-2 kg additional fat loss, primarily from subcutaneous stores in the lower abdomen and flanks. The effect on truly "stubborn" subcutaneous fat (which is resistant due to higher alpha-2 adrenergic receptor density) is unclear. AOD-9604 works through beta-3 receptors, and areas rich in alpha-2 receptors may be relatively resistant to its effects.
Visual changes: subtle improvement in abdominal definition, slightly more visible obliques, mild reduction in lower abdominal "pouch." These changes are visible to the individual but unlikely to be noticed by others.
For tracking changes accurately, take photos in identical lighting and posture every 2 weeks. Waist measurements and skinfold calipers provide more reliable data than the scale for individuals at lower body fat percentages.
What AOD-9604 Will Not Do
Setting boundaries on expectations prevents disappointment and inappropriate use.
AOD-9604 will not produce the 20-50 pound transformations that GLP-1 agonists produce. Those results require sustained caloric deficits of 500-750 calories per day driven by appetite suppression. AOD-9604 does not suppress appetite.
AOD-9604 will not overcome a caloric surplus. If you eat more calories than you burn, the free fatty acids released by AOD-9604 will simply be re-esterified and stored again. The compound mobilizes fat; it does not force the body to oxidize it. A caloric deficit is required for the mobilized fat to be burned as fuel.
AOD-9604 will not spot-reduce fat from specific areas. While beta-3 receptor density varies between fat depots (higher in visceral, moderate in abdominal subcutaneous, lower in gluteal-femoral), the peptide works systemically. You cannot target your belly specifically.
AOD-9604 will not build muscle. The fragment does not activate the growth hormone receptor or stimulate IGF-1 production. If muscle growth is a goal, consider GH secretagogues like ipamorelin or CJC-1295. For muscle-focused stacking, see the hexarelin dosage guide and the follistatin-344 dosage article. For skin and tissue-focused peptide dosing, see our GHK-Cu injection dosage guide.
Maximizing AOD-9604 Results: The Factors That Matter
AOD-9604 is an enhancer, not a replacement for the fundamentals. The users who report the best before-and-after outcomes share several habits that amplify the peptide's fat-mobilizing effect.
Caloric Deficit: The Non-Negotiable Foundation
AOD-9604 releases free fatty acids from adipocytes. If those fatty acids are not burned through a caloric deficit or exercise, they circulate briefly and are re-stored. The compound creates an opportunity for fat loss; the caloric deficit converts that opportunity into actual results.
A moderate deficit of 300-500 calories per day is sufficient. Extreme deficits (800-1,000 calories below maintenance) are unnecessary and counterproductive because they increase muscle catabolism and metabolic adaptation. AOD-9604 does not protect against the hormonal consequences of severe restriction the way GLP-1 agonists do.
Track your intake for at least the first 4 weeks to ensure you are consistently in deficit. The most common reason for AOD-9604 "not working" is that the user is not actually in a caloric deficit.
Fasted Exercise Timing
Injecting AOD-9604 30-45 minutes before fasted cardio creates a synergistic window. The peptide mobilizes fatty acids from adipocytes, and the exercise oxidizes those fatty acids before they can be re-esterified.
Low-to-moderate intensity cardio (brisk walking, cycling at conversation pace, light jogging) performed in this window maximizes fat oxidation because the exercise intensity keeps the body in a predominantly fat-burning metabolic state. High-intensity exercise shifts fuel utilization toward glycogen, reducing the advantage of the pre-mobilized fatty acids.
A practical protocol: wake, inject AOD-9604, wait 20 minutes, perform 30-45 minutes of moderate cardio, then eat breakfast. This approach stacks three fat-loss drivers (fasting, AOD-9604, exercise) into a single window.
Resistance Training and Protein Intake
While AOD-9604 appears to preserve lean mass based on animal data, combining it with resistance training and adequate protein provides an additional layer of protection. The animal studies showed preserved lean mass, but controlled human body composition data is not available.
Protein intake of 1.6-2.0 g/kg body weight during a caloric deficit is supported by extensive weight loss research for lean mass preservation (Wycherley et al., 2012). Resistance training 2-3 times per week with compound movements signals the body to retain muscle tissue while directing the caloric deficit toward fat stores.
This combination is especially important for AOD-9604 users because the compound does not stimulate growth hormone, IGF-1, or any anabolic pathway. Unlike a GH secretagogue stack that provides some inherent muscle-protective signaling, AOD-9604 offers fat loss only. You must supply the muscle-preservation stimulus through diet and training.
For comprehensive stacking protocols that combine AOD-9604 with muscle-preserving peptides, see our peptide stacking guide.
AOD-9604 Stacking: Combinations That Users Report
Because AOD-9604 targets a single pathway (beta-3 adrenergic lipolysis), many users combine it with peptides that address complementary mechanisms. The stacks below are commonly discussed in clinical practice and user communities. Evidence for these combinations comes from the individual compound data; no controlled trial has tested them together.
AOD-9604 + Ipamorelin (Fat Loss + GH Benefits)
This stack pairs AOD-9604's direct fat mobilization with ipamorelin's growth hormone release. Ipamorelin stimulates natural GH secretion, which improves sleep quality, supports recovery, and provides modest anabolic signaling to preserve lean mass during a caloric deficit.
Typical protocol: AOD-9604 300 mcg in the morning (fasted) + ipamorelin 200-300 mcg before bed. Both require fasted administration, so the timing is straightforward. The AOD-9604 handles morning fat mobilization; the ipamorelin handles nighttime GH pulsatility.
The theoretical advantage: ipamorelin compensates for AOD-9604's lack of anabolic signaling. While AOD-9604 mobilizes fat without preserving muscle, ipamorelin's GH elevation provides some muscle-protective effect. The combination addresses fat loss from two angles (direct lipolysis and metabolic rate support) while adding recovery benefits that pure AOD-9604 lacks.
Consult our peptide dosage chart for ipamorelin dose titration details.
AOD-9604 + BPC-157 (Fat Loss + Joint Health)
An emerging application for AOD-9604 is joint health. Preliminary research suggests the peptide stimulates proteoglycan synthesis in cartilage, which may benefit osteoarthritis (Metabolic Pharmaceuticals, 2010). Combining it with BPC-157, the most widely used healing peptide, creates a protocol that addresses both fat loss and tissue repair.
Typical protocol: AOD-9604 300 mcg daily + BPC-157 250-500 mcg daily. Both have excellent safety profiles with no known interactions. This stack is popular among users who are overweight and also dealing with joint issues that limit their exercise capacity. Reducing body fat decreases mechanical load on joints, while both peptides may support cartilage health.
For more on joint-focused peptide protocols, see our peptides for joint pain guide. For BPC-157 safety information, see BPC-157 side effects.
AOD-9604 + CJC-1295/Ipamorelin (Comprehensive Recomposition)
The triple stack of AOD-9604, CJC-1295 (DAC or no-DAC), and ipamorelin targets fat loss, GH elevation, and body recomposition simultaneously. CJC-1295 extends the GH release window beyond what ipamorelin alone provides, creating sustained overnight GH elevation that supports fat oxidation, muscle protein synthesis, and deep sleep.
Typical protocol: AOD-9604 300 mcg AM (fasted) + CJC-1295/Ipamorelin 100/200 mcg before bed. The AOD-9604 provides direct daytime fat mobilization. The CJC-1295/ipamorelin combination amplifies nighttime GH pulsatility for recovery and metabolic support.
This is the most comprehensive AOD-9604 stack, but it also represents the most complexity and cost. Use our peptide cost calculator to estimate monthly expenses before committing. For sourcing, see where to buy peptides in 2026.
Safety Profile and Side Effects
AOD-9604's safety profile is its strongest feature. The compound has more safety data than efficacy data, which is unusual for a research peptide.
Clinical trial safety data: In the Phase 2 trial, adverse events were comparable to placebo. No serious adverse events were attributed to AOD-9604. No dose-limiting toxicities were observed at any tested dose. The compound did not affect fasting glucose, insulin, HbA1c, IGF-1, cortisol, thyroid hormones, or liver enzymes (Thompson et al., 2004).
FDA GRAS determination (2007): The FDA reviewed AOD-9604's toxicology data and granted Generally Recognized As Safe status for oral use as a food ingredient. This determination was based on chronic toxicity studies, genotoxicity studies, and human clinical trial safety data. GRAS status does not equal drug approval, but it does indicate that the regulatory body considered the compound safe for human consumption at the doses tested.
Reported side effects from clinical practice:
| Side Effect | Frequency | Severity | Duration |
|---|---|---|---|
| Injection site redness | 5-15% | Mild | Minutes to hours |
| Mild headache | 5-10% | Mild | First few days |
| Lightheadedness (fasted) | 3-5% | Mild | During injection window |
| Stomach discomfort | 2-5% | Mild | Transient |
| Chest tightness/flushing | <2% | Mild | Minutes |
Contraindications: Pregnancy, breastfeeding, active cancer, and known hypersensitivity. While AOD-9604 does not stimulate cell proliferation (no IGF-1 elevation), the precautionary principle applies for active malignancies.
The regulatory landscape for research peptides has tightened. For current information on peptide availability and regulatory changes, see our FDA peptide crackdown 2026 analysis.
AOD-9604 vs HGH Fragment 176-191: Which Produces Better Before-and-After Results?
This is the most common comparison because both peptides share the same core sequence. AOD-9604 and HGH Fragment 176-191 both consist of amino acids 176-191 from human growth hormone. The only structural difference is AOD-9604's additional tyrosine residue at the N-terminal end.
| Feature | AOD-9604 | HGH Fragment 176-191 |
|---|---|---|
| Sequence | hGH 176-191 + Tyr | hGH 176-191 |
| Clinical trials | Phase 2 (humans) | None (humans) |
| FDA GRAS | Yes (2007) | No |
| Mechanism | Beta-3 lipolysis + anti-lipogenesis | Beta-3 lipolysis + anti-lipogenesis |
| Half-life | ~30 minutes | ~30 minutes |
| Dosing | 250-500 mcg/day | 250-500 mcg/day |
| Safety data | Extensive | Limited |
The tyrosine modification in AOD-9604 improves peptide stability and may slightly enhance lipolytic activity. Comparative studies in obese mice showed that AOD-9604 produced slightly greater fat reduction than the unmodified fragment at equivalent doses. However, both peptides were effective, and the difference was not large.
The practical advantage of AOD-9604 over HGH Fragment 176-191 is the safety data. AOD-9604 has human clinical trial data and FDA GRAS status. HGH Fragment 176-191 has no human trial data and relies entirely on animal studies and extrapolation from AOD-9604's human data.
For before-and-after outcomes, users report similar results from both compounds. The mechanism is effectively identical. If choosing between them, AOD-9604 has the stronger evidence base and regulatory standing.
For a detailed comparison of all fat-loss peptide options, see our best peptides for weight loss comprehensive guide.
Important Limitations and Warnings
Honesty about AOD-9604's limitations serves users better than hype. Several points deserve emphasis.
Limited human clinical data. The Phase 2 trial is the only controlled human efficacy study. The trial tested oral administration primarily, and no controlled study has tested subcutaneous injection in humans. All injectable dosing protocols are extrapolations from the oral trial, animal data, and clinical practice experience. The compound did not advance to Phase 3 because the efficacy was insufficient for drug approval.
Most before-and-after claims are unverified. Transformation photos shared online rarely include controlled conditions, verified dosing, or isolation of AOD-9604's contribution from diet and exercise changes. A person who starts AOD-9604 simultaneously with a new diet and exercise program cannot attribute their results solely to the peptide.
GRAS is not drug approval. The FDA's GRAS determination confirms safety for oral consumption as a food ingredient. It says nothing about efficacy for fat loss. Marketing AOD-9604 as "FDA approved" is misleading, and any vendor making that claim should be viewed skeptically.
AOD-9604 is not a substitute for GLP-1 agonists in clinical obesity. For individuals with BMI above 30 or BMI above 27 with comorbidities, FDA-approved GLP-1 receptor agonists (semaglutide, tirzepatide) have far stronger evidence and produce dramatically greater weight loss. AOD-9604's niche is as an adjunct for individuals seeking modest additional fat mobilization with minimal side effects, not as a primary obesity treatment.
Quality and sourcing concerns. As a research peptide, AOD-9604 is not manufactured under the same regulatory oversight as pharmaceutical products. Peptide purity, stability, and accurate dosing depend on the supplier. Third-party testing (HPLC purity, mass spectrometry) is essential. See our guide on where to buy peptides in 2026 for sourcing considerations.
This article is educational content for research purposes. AOD-9604 is not an approved pharmaceutical. Consult a healthcare provider before starting any peptide protocol. For a thorough understanding of peptide safety considerations, see our peptide safety guide.
Frequently Asked Questions
How much fat can you realistically lose with AOD-9604 in 12 weeks?
The Phase 2 clinical trial showed approximately 2-3 kg of additional fat loss compared to placebo over 12 weeks. This is on top of what diet and exercise alone would produce. At 500 mcg per day with a consistent caloric deficit, responders may see 1-2 inches of waist circumference reduction. These are modest but measurable results when combined with disciplined nutrition.
Does AOD-9604 suppress appetite like semaglutide or tirzepatide?
No. AOD-9604 works entirely at the fat cell level through beta-3 adrenergic receptor activation. It does not interact with GLP-1 receptors, does not slow gastric emptying, and has no effect on hunger or satiety signals in the brain. You will eat the same amount on AOD-9604 as you would without it. A caloric deficit must come from conscious dietary choices, not peptide-driven appetite suppression.
Is AOD-9604 FDA approved for weight loss?
No. AOD-9604 received FDA GRAS (Generally Recognized As Safe) status as a food ingredient in 2007, but this is not drug approval. The compound was never approved as a pharmaceutical treatment for obesity because the Phase 2 clinical trial results, while showing safety and modest efficacy, did not meet the threshold for drug registration. GRAS confirms safety, not effectiveness as a weight loss drug.
Can I take AOD-9604 without exercising or dieting?
You can, but results will be negligible. AOD-9604 mobilizes fat by releasing free fatty acids from adipocytes. Without a caloric deficit to force the body to oxidize those fatty acids as fuel, they circulate briefly and are re-stored. The clinical trial participants were on controlled diets. Using AOD-9604 without dietary modification is unlikely to produce noticeable before-and-after changes.
What is the difference between AOD-9604 and HGH Fragment 176-191?
Both peptides share the same amino acid sequence from human growth hormone (positions 176-191). AOD-9604 has an additional tyrosine residue added at the N-terminus, which improves stability and may slightly enhance lipolytic activity. AOD-9604 has more clinical data behind it, including human trials and FDA GRAS status. HGH Fragment 176-191 has no published human trial data. Users report similar fat loss results from both.
Does AOD-9604 affect blood sugar, insulin, or growth hormone levels?
No. Multiple human and animal studies confirm that AOD-9604 has zero impact on fasting glucose, fasting insulin, HbA1c, IGF-1, cortisol, or thyroid hormones. The fragment does not bind the growth hormone receptor or activate the JAK2-STAT5 signaling pathway. This clean endocrine profile is the peptide's primary safety advantage over full HGH therapy or GH secretagogues.
How long should I run an AOD-9604 cycle?
The clinical trial tested 12 weeks. Most practitioners recommend 12-16 week cycles followed by a 2-4 week assessment break. AOD-9604 does not cause receptor desensitization or hormonal suppression, so the break is precautionary rather than biologically required. Some users run it continuously for 6 months without reported issues, but this exceeds the controlled evidence base. Reassess your results at week 8 to determine if continuing is worthwhile.
When is the best time to inject AOD-9604?
Morning, on a completely empty stomach. AOD-9604's lipolytic activity is suppressed by insulin, so fasted administration is essential. Inject upon waking, wait 20-30 minutes, then eat breakfast. For split-dose protocols (250 mcg twice daily), take the second dose at least 3 hours after your last meal. Injecting 30-45 minutes before fasted cardio may maximize fat oxidation during the exercise window.
The Bottom Line
AOD-9604 before-and-after results tell a straightforward story: modest additional fat loss with an exceptionally clean safety profile. The Phase 2 clinical trial showed roughly 2-3 kg more fat loss than placebo over 12 weeks, with no impact on blood sugar, insulin, IGF-1, or any hormonal marker. The compound received FDA GRAS status for safety but was never approved as a drug because efficacy was insufficient for pharmaceutical registration.
The honest assessment is that AOD-9604 is a mild fat-loss enhancer, not a transformation agent. It works best for individuals who already control their diet, exercise consistently, and want a marginal additional push in fat mobilization without gastrointestinal side effects or hormonal disruption. If you need dramatic weight loss, GLP-1 agonists like semaglutide or tirzepatide produce 5-10 times greater results through an entirely different mechanism.
The limited human evidence is the elephant in the room. Animal data is strong. Safety data is excellent. But controlled human efficacy data is sparse, and the single Phase 2 trial tested oral administration rather than the subcutaneous injection most users employ today. Every timeline beyond 12 weeks, every injectable dosing recommendation, and every stacking protocol is an extrapolation rather than a validated finding.
Use our peptide reconstitution calculator for preparation and our peptide cost calculator to estimate monthly expenses. For sourcing, see where to buy peptides in 2026.
Related articles: - Best Peptides for Weight Loss - comprehensive comparison of all fat loss peptides - Does Tirzepatide Burn Fat? - how GLP-1/GIP dual agonism compares to direct lipolysis - Retatrutide vs Tirzepatide - the triple agonist achieving 24.2% weight loss - How Does Retatrutide Work? - glucagon receptor activation and hepatic thermogenesis - Peptide Dosage Chart - complete dosing reference for all peptides - Peptide Safety Guide - side effects, interactions, and risk management
Explore all peptide profiles and tools at PeptidesExplorer.
Helpful Tools
Related Articles
Sermorelin for Fat Loss: Dosing & Data
Sermorelin stimulates natural GH release to enhance fat oxidation. Dosing protocols (100-300mcg/day), cycling, and CJC-1295 comparison.
Does Tirzepatide Burn Fat?
Tirzepatide produced 33.9% fat mass loss in SURMOUNT-1. Learn how GLP-1/GIP dual agonism drives fat loss, body composition data, and how to protect lean mass.
9 Best Peptides for Weight Loss (2026)
9 best peptides for weight loss ranked: semaglutide, tirzepatide, retatrutide, AOD-9604, CJC-1295. Clinical trial data, dosages, and expected results.
5-Amino-1MQ Dosage: Protocols, Cycling, and Safety
5-Amino-1MQ dosage guide: 50-150 mg/day oral protocols, NNMT inhibition mechanism, cycle length, stacking options, and PubMed-cited research.