You searched "is GHK-Cu FDA-approved" because you want a straight answer before you buy a copper peptide serum or consider injectable therapy. No, GHK-Cu is not FDA-approved as a drug. The FDA has never granted New Drug Application (NDA) approval for GHK-Cu in any form, for any indication. It remains classified as a cosmetic ingredient when sold in topical products and as a bulk drug substance eligible for compounding when prescribed by a licensed physician (FDA CDER, Bulk Drug Substances, 2024).
| Quick Reference | Details |
|---|---|
| FDA-approved as a drug? | No |
| Legal in cosmetics? | Yes, as "Copper Tripeptide-1" (INCI) |
| Legal for compounding? | Yes, Category 1 since Feb 2026 |
| Prescription required for injections? | Yes |
| Number of Phase III clinical trials completed | Zero |
| Estimated cost for FDA drug approval | $1-2 billion |
| Years of published research | 50+ (since 1973) |
| Total PubMed studies | 200+ |
This distinction matters. "Not FDA-approved" does not mean "unsafe" or "illegal." It means no pharmaceutical company has invested the $1-2 billion required to run Phase III clinical trials and submit an NDA. The reasons are economic, not scientific. This guide explains exactly what GHK-Cu is, how the FDA classifies it, what changed in February 2026, and what your options are for legal access.
Get your custom peptide protocol:
- Tailored to your body and goals
- Precise dosing and cycle length
- Safe stacking combinations
- Backed by peer-reviewed studies
- Ready in under 2 minutes
What Is GHK-Cu?
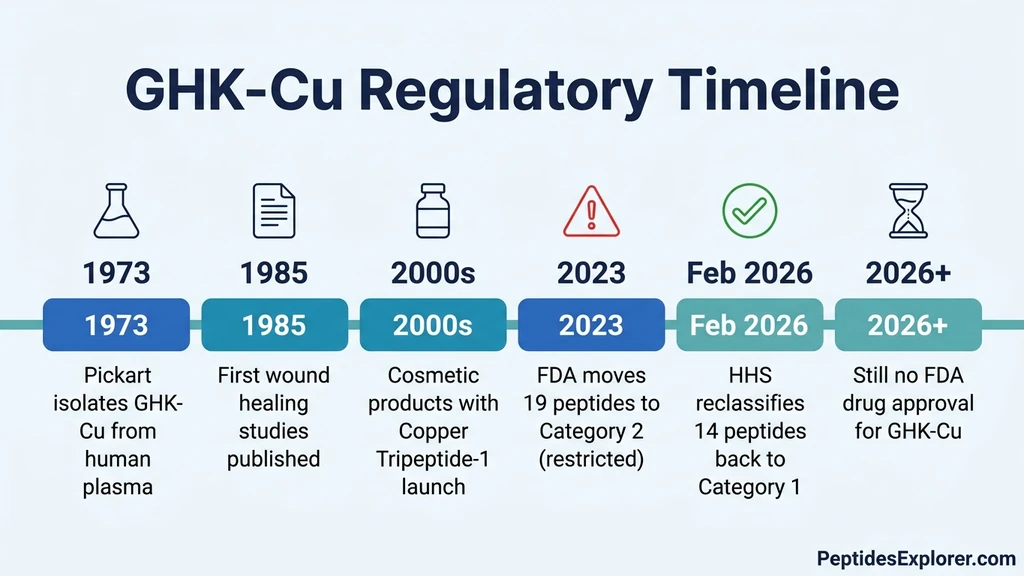
GHK-Cu (glycyl-L-histidyl-L-lysine copper complex) is a naturally occurring tripeptide bound to a copper(II) ion. Your body produces it. It circulates in human plasma at approximately 200 ng/mL at age 20 and declines to roughly 80 ng/mL by age 60. Loren Pickart first isolated GHK-Cu from human blood plasma in 1973 while studying age-related changes in liver cell function (Pickart & Thaler, *Nature New Biology*, 1973).
The peptide modulates over 4,000 human genes involved in tissue repair, inflammation, and antioxidant defense. It promotes collagen synthesis, accelerates wound closure, stimulates hair follicle growth, and reduces oxidative damage. For a complete breakdown, see our GHK-Cu benefits guide.
What makes GHK-Cu unusual in the peptide world is its dual identity. It exists simultaneously as a cosmetic ingredient (sold in serums and creams under the INCI name "Copper Tripeptide-1") and as a research compound used in injectable form. These two paths have completely different regulatory frameworks.
FDA Approval: What It Actually Means
Before analyzing GHK-Cu's regulatory status, you need to understand what "FDA-approved" actually requires. The phrase carries specific legal meaning that most consumers misunderstand.
The FDA Drug Approval Process
FDA drug approval follows a rigid sequence that typically spans 10-15 years and costs $1-2 billion:
| Phase | Purpose | Duration | Typical Cost |
|---|---|---|---|
| Preclinical | Animal studies, toxicology | 1-3 years | $10-50M |
| IND Application | Permission to test in humans | 1 month review | $1-5M |
| Phase I | Safety in healthy volunteers (20-80 people) | 1-2 years | $15-30M |
| Phase II | Efficacy and dose-finding (100-300 people) | 2-3 years | $20-50M |
| Phase III | Large-scale efficacy (1,000-3,000 people) | 3-4 years | $100M-1B |
| NDA Review | FDA evaluates full data package | 6-12 months | $5-10M |
| Post-market (Phase IV) | Ongoing safety surveillance | Ongoing | Varies |
GHK-Cu has never entered this pipeline. No Investigational New Drug (IND) application has been filed. No Phase I trial for injectable GHK-Cu has been registered with the FDA. The compound exists entirely outside the pharmaceutical approval framework.
This is not unusual. The vast majority of naturally occurring peptides in your body have never gone through FDA drug approval. Insulin required approval because pharmaceutical companies manufactured synthetic versions and sold them as drugs. GHK-Cu has not followed that path.
Why "Not FDA-Approved" Does Not Mean "Unsafe"
The FDA approval process is designed to evaluate manufactured pharmaceuticals. It verifies safety and efficacy for specific medical claims. Absence of approval means one of two things: either the compound failed trials, or no one ran them. For GHK-Cu, it is the second case.
Many substances you consume daily are not FDA-approved as drugs. Vitamin D, omega-3 fatty acids, and melatonin are all sold as dietary supplements without NDA approval. The FDA regulates them under different frameworks (DSHEA for supplements, FDCA for cosmetics) rather than through the drug approval pipeline.
GHK-Cu's safety profile after 50 years of research and commercial cosmetic use is well-documented. No serious adverse events have been reported in published literature. The compound is identical to what your body produces naturally. For injectable use, the risks relate to compounding quality, sterility, and administration technique rather than the peptide itself. See our GHK-Cu side effects guide for the complete safety profile.
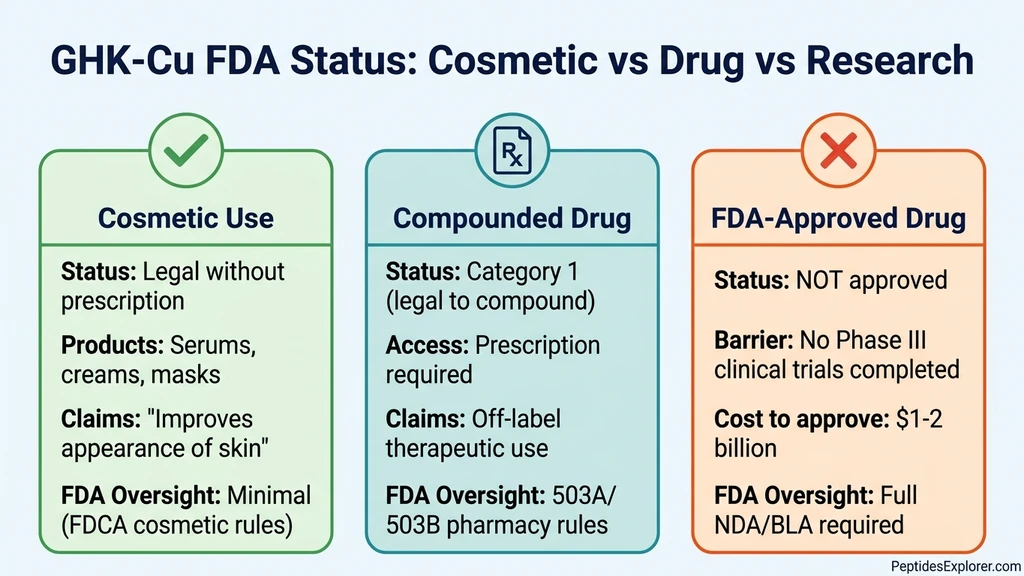
GHK-Cu's Three Regulatory Categories
GHK-Cu occupies three distinct regulatory spaces depending on how it is formulated, marketed, and sold. Each category has different rules, different levels of FDA oversight, and different legal requirements.
1. Cosmetic Ingredient (Copper Tripeptide-1)
When GHK-Cu appears in serums, creams, masks, and other topical products, it is regulated as a cosmetic under the Federal Food, Drug, and Cosmetic Act (FDCA). No FDA approval is required to sell cosmetics. No prescription is needed to buy them.
The key legal boundary is the marketing claims. A cosmetic product can claim to "improve the appearance of fine lines" or "support skin radiance." It cannot claim to "treat wrinkles," "heal wounds," or "reverse aging." The moment a manufacturer makes a therapeutic claim, the product becomes an unapproved drug under FDA rules.
Copper Tripeptide-1 is the International Nomenclature of Cosmetic Ingredients (INCI) name for GHK-Cu. It appears on ingredient lists of products from brands like The Ordinary, NIOD, and dozens of professional skincare lines. The Cosmetic Ingredient Review (CIR) Expert Panel has assessed copper peptides and found them safe for cosmetic use at concentrations typically used in formulations.
This is the most accessible form of GHK-Cu. You can buy it today without a prescription, without a physician, and without any FDA involvement. For topical application methods, see our GHK-Cu microneedling guide and what peptides do for skin.
2. Compounded Drug (Category 1 Bulk Drug Substance)
Injectable GHK-Cu falls under a different framework. When a physician prescribes GHK-Cu and a compounding pharmacy prepares it, the product is regulated under Section 503A or 503B of the FDCA. This is not FDA drug approval. It is a legal pathway for pharmacists to prepare customized medications that are not commercially available as approved drugs.
What changed in February 2026: On February 27, 2026, HHS Secretary Robert F. Kennedy Jr. announced the reclassification of approximately 14 peptides from Category 2 (restricted) back to Category 1 (eligible for compounding). GHK-Cu was among the peptides affected. Previously, in late 2023, the FDA had moved 19 peptides to its Category 2 list, effectively prohibiting compounding pharmacies from preparing them.
Category 1 status means: - Licensed 503A pharmacies can compound GHK-Cu with a valid prescription - Licensed 503B outsourcing facilities can produce it in larger batches - A prescriber-patient relationship is required - The compounding pharmacy must follow current Good Manufacturing Practices (cGMP) - The product is considered an off-label therapy, not an FDA-approved drug
For the full regulatory landscape, see our FDA peptide crackdown 2026 analysis and are peptides legal guide.
3. Research Chemical
GHK-Cu is also sold by research chemical suppliers labeled "for research purposes only" or "not for human consumption." This market operates in a gray area. The compounds are legal to purchase for laboratory research. They are not legal to self-administer, though enforcement is minimal.
Research-grade GHK-Cu lacks the quality controls of pharmaceutical compounding. Purity verification, endotoxin testing, and sterility assurance vary widely between suppliers. The cost is significantly lower than compounded prescriptions, which is why some users choose this route despite the risks.
The FDA does not regulate research chemicals sold for non-human use. However, if a company markets a research chemical with implicit human use instructions (dosage guidelines, injection protocols), the FDA can take enforcement action for selling an unapproved drug.
Why GHK-Cu Will Likely Never Get FDA Drug Approval
The absence of FDA approval for GHK-Cu is not a scientific failure. It is an economic reality. Understanding the structural barriers explains why this peptide with 50 years of research behind it remains outside the pharmaceutical approval system.

The Patent Problem
GHK-Cu is a naturally occurring human peptide. You cannot patent a natural substance in the United States. Without patent protection, a pharmaceutical company that spends $1-2 billion on clinical trials cannot prevent competitors from selling generic versions the day after approval. The return on investment is effectively zero.
This is the single biggest reason GHK-Cu has no FDA approval. The drug approval process was designed for novel synthetic molecules that companies can patent, price at a premium, and sell exclusively for 20 years. Natural peptides do not fit this model.
Pharmaceutical companies can patent specific formulations, delivery mechanisms, or modified analogs of GHK-Cu. Some have tried. ProCyte Corporation held patents on copper peptide formulations in the 1990s and early 2000s. But a formulation patent is narrower than a composition-of-matter patent. Competitors can reformulate and sell their own versions, limiting the market exclusivity that justifies billion-dollar trial investments.
The Indication Problem
FDA approval is granted for specific indications: "for the treatment of [disease X]." GHK-Cu's research spans wound healing, skin aging, hair loss, inflammation, neurodegeneration, and cancer gene modulation. No single indication has enough commercial potential to justify the trial cost, and running separate trials for each indication multiplies the investment.
Contrast this with tirzepatide, which received FDA approval for type 2 diabetes (a massive market) with a clear, measurable endpoint (HbA1c reduction). The business case was obvious. For GHK-Cu applied to wound healing in a small patient population, the math does not work.
The Loren Pickart Factor
Loren Pickart, the biochemist who discovered GHK-Cu in 1973, spent his career in academic and independent research rather than pharmaceutical development. His work produced over 100 publications documenting GHK-Cu's mechanisms and effects. He co-founded Skin Biology, a company that sold copper peptide skincare products commercially.
Pickart's research established the scientific foundation: GHK-Cu stimulates collagen I and III synthesis, accelerates wound closure, upregulates decorin (an anti-cancer proteoglycan), modulates TGF-beta signaling, and resets gene expression patterns toward youthful profiles (Pickart et al., *Int J Mol Sci*, 2018). His 2012 genomic analysis using the Broad Institute Connectivity Map showed that GHK modulates 4,000+ genes (Pickart et al., *BioMed Res Int*, 2012).
What Pickart never had was the financial backing to run Phase III clinical trials. Academic researchers do not have $500 million to spend on a single randomized controlled trial. Pharmaceutical companies did not see a patentable product worth investing in. The result: strong basic science, robust mechanistic data, and zero clinical trials designed for FDA submission.
What the Research Actually Shows
GHK-Cu's evidence base is substantial for a compound without FDA approval. Over 200 published studies spanning five decades document its biological activity. The limitation is not the volume of research but the type: most studies are in vitro (cell culture) or animal models, with only small human studies for wound healing and cosmetic applications.
Wound Healing (Strongest Evidence)
The earliest clinical application of GHK-Cu was wound healing. Pickart and colleagues demonstrated in the 1980s that copper peptides accelerate wound closure, improve the take of transplanted skin grafts, and reduce scar formation.
A controlled study of copper peptide-containing wound dressings showed statistically significant improvement in healing rates compared to control dressings. Patients with diabetic ulcers, surgical wounds, and burn injuries showed faster epithelialization and reduced inflammation.
These studies were small (typically fewer than 50 patients) and predated modern clinical trial standards. They demonstrate biological plausibility and preliminary efficacy but do not meet the evidentiary threshold for FDA drug approval, which requires large, multi-center, randomized, double-blind, placebo-controlled Phase III trials.
For injectable applications and dosing protocols, see our GHK-Cu injection dosage guide.
Skin Rejuvenation (Moderate Evidence)
Cosmetic studies of topical copper peptides have shown measurable improvements in skin parameters. A 12-week facial study demonstrated increased skin thickness, reduced fine lines, and improved skin density as measured by ultrasound imaging. Copper peptide formulations performed comparably to retinol and vitamin C in head-to-head cosmetic trials.
These results support the cosmetic claims that manufacturers make on product labels. They do not, however, constitute evidence for a drug claim. The distinction matters: "improves appearance of wrinkles" is a cosmetic claim that does not require FDA approval. "Treats photoaging" is a drug claim that does.
GHK-Cu's mechanisms for skin improvement are well-characterized at the cellular level. It stimulates fibroblast collagen synthesis, increases glycosaminoglycan production (the "moisture" matrix), promotes angiogenesis (new blood vessel formation), and reduces oxidative damage via superoxide dismutase (SOD) upregulation. The mechanistic data is strong. The clinical trial data is thin.
Hair Growth (Emerging Evidence)
GHK-Cu promotes hair growth through multiple pathways: Wnt/beta-catenin signaling activation (the master switch for hair follicle cycling), VEGF upregulation (blood supply to follicles), enlarged follicle size, and copper-mediated type 1 5-alpha reductase inhibition that reduces local DHT production (Sugimoto et al., 1995).
No randomized controlled trial has tested injectable or topical GHK-Cu specifically for androgenetic alopecia as a primary endpoint. The evidence comes from mechanistic studies, in vitro hair follicle models, and observational reports. For a deep dive into the hair growth mechanisms, see GHK-Cu hair growth and does GHK-Cu block DHT.
Gene Modulation and Anti-Aging (Preclinical)
The 2012 Broad Institute Connectivity Map analysis remains one of the most striking findings in GHK-Cu research. Pickart and colleagues showed that GHK modulates the expression of 4,000+ human genes, shifting expression patterns from "aged" toward "youthful" profiles. Key gene categories affected include:
- DNA repair genes: Upregulated, suggesting enhanced genomic maintenance
- Antioxidant genes: Increased SOD and other protective enzymes
- Anti-cancer genes: Upregulated caspase and growth regulatory genes
- Inflammatory genes: Downregulated NF-kB and TNF-alpha pathways
- Tissue remodeling genes: Increased collagen, elastin, and matrix metalloproteinase regulators
This data is from gene expression microarray analysis, not clinical outcomes. Gene modulation does not automatically translate to measurable health benefits in living humans. The gap between "changes gene expression in cell culture" and "prevents aging in a Phase III trial" is enormous. No one has bridged that gap for GHK-Cu, and the patent economics ensure that no one is likely to try.
How to Access GHK-Cu Legally in 2026
Despite the absence of FDA drug approval, GHK-Cu is legal to obtain through multiple channels in the United States. The route you choose determines the quality, cost, and level of medical oversight.
Topical Products (No Prescription Required)
Copper Tripeptide-1 is available in over-the-counter skincare products from dozens of manufacturers. Concentrations typically range from 0.01% to 1%. Products include serums, eye creams, moisturizers, and professional-grade formulations used in aesthetic clinics.
Popular topical options include buffered copper peptide solutions (often sold as "GHK-Cu serum"), combination products with hyaluronic acid and niacinamide, and professional microneedling solutions. Prices range from $15 for basic formulations to $150+ for high-concentration professional products.
Topical application does not require a prescription, a physician consultation, or any regulatory approval. This is the simplest and most accessible route. For microneedling protocols with GHK-Cu, see our GHK-Cu microneedling guide.
Compounded Injections (Prescription Required)
Since the February 2026 reclassification, compounding pharmacies can again legally prepare injectable GHK-Cu with a valid prescription. The process works as follows:
- 1.Physician consultation: A licensed provider evaluates your medical history and determines if GHK-Cu is appropriate
- 2.Prescription: The provider writes a prescription specifying the dose, concentration, and quantity
- 3.Compounding: A licensed 503A or 503B pharmacy prepares the injectable formulation
- 4.Delivery: The compounded product is shipped directly to you or the prescribing clinic
Costs for compounded injectable GHK-Cu typically range from $80-200 per vial (5-10mg), depending on the pharmacy and concentration. Telehealth peptide clinics have streamlined this process, offering consultations, prescriptions, and pharmacy coordination in a single platform.
For reconstitution instructions if you receive lyophilized powder, use our peptide reconstitution calculator. For injection technique and site selection, see GHK-Cu nasal spray for alternative delivery methods.
What About Other Peptides?
GHK-Cu's regulatory status mirrors that of most therapeutic peptides. BPC-157 and TB-500 share the same pattern: extensive preclinical research, no FDA drug approval, legal access through compounding pharmacies with a prescription. The February 2026 reclassification affected all three compounds along with 11 others.
The broader peptide regulatory landscape is evolving. For a comprehensive analysis of which peptides are legal and how to access them, see our are peptides legal guide and FDA peptide crackdown 2026 timeline.
Cosmetic vs Drug Claims: The Legal Line
The distinction between a cosmetic and a drug under FDA rules is not based on the ingredient. It is based on the intended use, which the FDA determines primarily from marketing claims. The same molecule (GHK-Cu) can be a legal cosmetic or an illegal unapproved drug depending on what the seller says about it.
Permitted Cosmetic Claims
Under the FDCA, a cosmetic is defined as an article "intended to be applied to the human body for cleansing, beautifying, promoting attractiveness, or altering the appearance." GHK-Cu products can legally claim:
- "Improves the appearance of fine lines and wrinkles"
- "Supports skin radiance and firmness"
- "Helps maintain youthful-looking skin"
- "Promotes a healthy, even skin tone"
These are appearance-based claims. They describe what the product looks like on the surface, not what it does at the cellular level.
Prohibited Drug Claims (Without FDA Approval)
A drug is defined under the FDCA as an article "intended for use in the diagnosis, cure, mitigation, treatment, or prevention of disease" or "intended to affect the structure or any function of the body." GHK-Cu products cannot legally claim:
- "Stimulates collagen production" (affects body structure)
- "Treats wrinkles" (implies treatment of a condition)
- "Heals wounds" (therapeutic claim)
- "Reverses skin aging" (affects body function)
- "Blocks DHT" (pharmacological action)
This creates an irony. GHK-Cu genuinely does stimulate collagen production. The published research confirms it. But stating that on a product label transforms the cosmetic into an unapproved drug, triggering potential FDA enforcement. Sellers must describe what GHK-Cu looks like it does ("improves appearance of firmness") rather than what it biochemically does ("increases type I collagen synthesis").
For the science behind GHK-Cu's actual mechanisms, see our GHK-Cu benefits review.
GHK-Cu Compared to FDA-Approved Peptides
Several peptides have achieved full FDA drug approval, providing useful context for understanding why GHK-Cu has not.
Peptides That Did Get FDA Approval
| Peptide | Brand Name | FDA Approval | Indication | Sponsor |
|---|---|---|---|---|
| Semaglutide | Ozempic, Wegovy | 2017/2021 | Diabetes, Obesity | Novo Nordisk |
| Tirzepatide | Mounjaro, Zepbound | 2022/2023 | Diabetes, Obesity | Eli Lilly |
| Tesamorelin | Egrifta | 2010 | HIV lipodystrophy | Theratechnologies |
| Bremelanotide | Vyleesi | 2019 | Hypoactive sexual desire | Palatin |
Every peptide on this list shares three characteristics that GHK-Cu lacks:
- 1.Patentability: All are synthetic or modified peptides that companies could patent, protecting their investment
- 2.Large market: Each targets a condition affecting millions of patients, making the $1-2 billion investment recoverable
- 3.Corporate sponsor: Major pharmaceutical companies (Novo Nordisk, Eli Lilly) with the capital and infrastructure to run global Phase III trials
GHK-Cu has none of these advantages. It is a natural, unpatentable peptide with fragmented potential indications and no pharmaceutical sponsor.
What This Means for Your Decision
The comparison reveals that FDA approval is as much an economic filter as a scientific one. GHK-Cu is not less studied than bremelanotide was before its approval. It simply lacks the market structure that incentivizes a company to spend $1 billion proving what academic research already suggests.
This does not mean you should treat GHK-Cu and FDA-approved drugs as equivalent. FDA approval provides standardized dosing, manufacturing quality assurance, adverse event monitoring, and legal accountability. Compounded and research-grade GHK-Cu lacks these safeguards. The tradeoff is access to a compound with strong preclinical data but without the gold-standard clinical validation that the pharmaceutical approval process provides.
For dosage guidance compiled from published research, see our GHK-Cu injection dosage guide.
Safety Profile Without FDA Oversight
The absence of FDA drug approval means GHK-Cu lacks the post-market surveillance infrastructure that monitors approved drugs. However, its safety profile is well-characterized through other channels.
50 Years of Topical Use
Copper peptides have been used in commercial skincare products since the late 1990s. Millions of consumers have applied GHK-Cu topically without reports of serious adverse events reaching the published literature. The Cosmetic Ingredient Review Expert Panel has assessed copper peptides and determined them safe for cosmetic use.
Common topical side effects are limited to mild irritation in sensitive individuals, typically at higher concentrations. No systemic toxicity has been reported from topical application. For complete topical safety data, see our GHK-Cu side effects guide.
Injectable Safety Considerations
Injectable GHK-Cu introduces additional variables that topical use does not. The primary risks are not from the peptide itself but from the compounding and administration process:
- Sterility: Contaminated products can cause injection site infections or systemic sepsis
- Purity: Impurities in poorly manufactured peptides can trigger immune reactions
- Endotoxin levels: Bacterial endotoxins in injectable products cause fever and inflammation
- Injection technique: Improper subcutaneous injection can cause tissue damage or abscess
These risks are mitigated by using licensed compounding pharmacies (503A or 503B) that follow cGMP standards, proper reconstitution technique, and sterile injection protocols. Research-grade peptides from unregulated suppliers carry substantially higher risk because they lack pharmaceutical-grade quality controls.
GHK-Cu itself has low inherent toxicity. The lethal dose in animal studies is orders of magnitude above therapeutic doses. No organ toxicity has been observed at standard doses in published research. For concerns about specific organ systems, see does GHK-Cu cause liver damage and does GHK-Cu cause hair loss.
The February 2026 Reclassification: What Actually Changed
The February 2026 announcement by HHS Secretary Robert F. Kennedy Jr. was the most significant regulatory shift for peptide therapy in a decade. Understanding exactly what changed, and what did not, prevents common misconceptions.
Before: The 2023 Category 2 Restriction
In late 2023, the FDA placed 19 widely used peptides on its Category 2 list of bulk drug substances that "raise significant safety or effectiveness concerns." Category 2 effectively banned compounding pharmacies from preparing these peptides. The list included GHK-Cu, BPC-157, TB-500, and other popular therapeutic peptides.
The FDA cited insufficient safety data and quality concerns in compounded formulations. Critics argued that the move was influenced by pharmaceutical industry pressure, as compounded peptides competed with expensive branded drugs. The reclassification disrupted access for thousands of patients receiving peptide therapy through licensed clinics and physicians.
After: The 2026 Category 1 Restoration
The February 2026 reclassification moved approximately 14 of the 19 restricted peptides back to Category 1. For GHK-Cu, this means:
- Legal to compound again: Licensed pharmacies can prepare injectable GHK-Cu with a valid prescription
- Not FDA-approved: The reclassification did not grant drug approval. No clinical trial data was submitted. No NDA was reviewed
- Still off-label: Physicians prescribing GHK-Cu are doing so off-label, meaning for uses not reviewed by the FDA
- Quality requirements remain: Compounding pharmacies must still follow cGMP and state pharmacy board regulations
The reclassification restored the status quo that existed before late 2023. It did not create new legal status for GHK-Cu. It reversed a restriction that many practitioners and patients considered overreach. For the complete timeline and affected peptides, see our FDA peptide crackdown 2026 analysis.
Could GHK-Cu Ever Get FDA Approval?
The structural barriers to FDA approval for GHK-Cu are real but not absolute. Several scenarios could change the equation, though none is likely in the near term.
Possible Pathways
Modified analog approach: A pharmaceutical company could develop a patentable modified version of GHK-Cu (altered amino acid sequence, novel delivery system, or prodrug formulation) that retains the biological activity while qualifying for composition-of-matter patent protection. This is the most realistic pathway but produces a different molecule, not GHK-Cu itself.
Orphan drug designation: If GHK-Cu were targeted at a rare disease (affecting fewer than 200,000 Americans), the sponsor would receive 7 years of market exclusivity, reduced FDA fees, and tax credits. This partially solves the patent problem. Potential orphan indications might include rare wound-healing disorders or specific dermatological conditions.
505(b)(2) pathway: This regulatory pathway allows applicants to rely on published literature and studies not conducted by the applicant. It reduces the clinical trial burden and cost. A 505(b)(2) application for GHK-Cu could leverage the existing 200+ published studies, potentially cutting the timeline and cost by 50% or more.
Academic-sponsored trial: A university or research institution could run a smaller, publicly funded clinical trial for a specific indication. This would not produce commercial FDA approval but could generate the Phase II data that might attract a pharmaceutical partner.
Realistic Outlook
None of these pathways is currently being pursued for GHK-Cu. No IND application is on file. No pharmaceutical company has announced plans to develop GHK-Cu as a drug. The economic fundamentals have not changed since Pickart's original research in the 1970s.
The most likely future for GHK-Cu is continued dual existence: a widely available cosmetic ingredient and a physician-prescribed compounded therapeutic, with the regulatory gray area persisting indefinitely. The 2026 reclassification solidified this middle ground rather than pushing toward either full approval or full restriction.
Frequently Asked Questions
Is GHK-Cu FDA-approved?
No. GHK-Cu has never received FDA drug approval for any indication. No New Drug Application (NDA) has been submitted, and no Phase III clinical trial has been completed. It is legal as a cosmetic ingredient (Copper Tripeptide-1) and as a compounded drug prescribed by a licensed physician. For the full GHK-Cu profile, see our peptide database.
Is GHK-Cu legal to buy?
Yes. Topical GHK-Cu products (serums, creams) are legal to purchase without a prescription as cosmetics. Injectable GHK-Cu requires a prescription from a licensed provider and must be prepared by a licensed compounding pharmacy. Research-grade GHK-Cu can be purchased legally for laboratory use. For the complete legal framework, see our are peptides legal guide.
Why isn't GHK-Cu FDA-approved if there are 200+ studies?
The barrier is economic, not scientific. FDA drug approval costs $1-2 billion and requires Phase III clinical trials. GHK-Cu is a naturally occurring peptide that cannot be patented, meaning no company can recoup its investment through exclusive sales. Published studies are predominantly in vitro and animal models, not the large randomized controlled trials the FDA requires. Learn more about the research in our GHK-Cu benefits analysis.
What changed with the 2026 FDA peptide reclassification?
In February 2026, HHS Secretary RFK Jr. moved approximately 14 peptides (including GHK-Cu) from Category 2 (restricted) back to Category 1 (eligible for compounding). This restored legal compounding access through licensed pharmacies with a prescription. It did not grant FDA drug approval. For the full breakdown, see our FDA peptide crackdown 2026 timeline.
Can I get injectable GHK-Cu with a prescription?
Yes. Since the February 2026 reclassification, licensed compounding pharmacies can prepare injectable GHK-Cu with a valid prescription from a licensed provider. Telehealth peptide clinics offer consultations and prescriptions remotely. For dosing guidance, see our GHK-Cu injection dosage guide, and for preparation instructions, use our peptide reconstitution calculator.
Is GHK-Cu safe without FDA approval?
GHK-Cu has a strong safety profile based on 50 years of research and commercial cosmetic use. No serious adverse events have been reported in published literature. The compound is identical to a peptide your body produces naturally. Injectable use introduces compounding and sterility risks unrelated to the peptide itself. Use licensed 503A or 503B pharmacies for injectable formulations. See our GHK-Cu side effects guide for the full safety profile.
What is the difference between cosmetic GHK-Cu and injectable GHK-Cu?
The molecule is identical. The difference is formulation, delivery method, and regulatory classification. Cosmetic products (serums, creams) are applied topically, regulated as cosmetics, and sold without prescription. Injectable formulations are prepared by compounding pharmacies, regulated as compounded drugs, and require a prescription. Injectable delivery achieves higher systemic bioavailability. For delivery method comparisons, see our GHK-Cu nasal spray guide.
Could GHK-Cu get FDA approval in the future?
It is theoretically possible but unlikely. Potential pathways include a modified patentable analog, orphan drug designation for a rare disease indication, or a 505(b)(2) application leveraging existing published literature. No pharmaceutical company has announced plans to pursue FDA approval for GHK-Cu. The economic fundamentals (unpatentable natural peptide, fragmented indications) have not changed since its discovery in 1973.
Are any copper peptides FDA-approved?
No copper peptide has received FDA drug approval for any indication. Copper peptides are widely used in FDA-regulated cosmetics under the INCI name Copper Tripeptide-1, but cosmetic regulation is separate from drug approval. The Cosmetic Ingredient Review Expert Panel has assessed copper peptides as safe for cosmetic use. For copper peptide skincare science, see what do peptides do for skin.
How does GHK-Cu compare to FDA-approved peptides like semaglutide?
Semaglutide (Ozempic, Wegovy) received FDA approval because Novo Nordisk invested billions in Phase III trials, the molecule is synthetic and patentable, and it targets a massive market (obesity, diabetes). GHK-Cu lacks all three advantages: it is natural and unpatentable, no company has funded large trials, and its indications (wound healing, skin rejuvenation) represent smaller markets. The scientific evidence for GHK-Cu is strong at the preclinical level but has not been translated into the clinical trial format the FDA requires.
The Bottom Line
GHK-Cu is not FDA-approved as a drug, and it probably never will be. The barrier is financial, not scientific. Fifty years of research, 200+ published studies, and 4,000+ modulated genes have established GHK-Cu as one of the most thoroughly characterized peptides in the biomedical literature. What is missing is a $1-2 billion investment in Phase III clinical trials that no company will make for an unpatentable natural peptide.
Your legal options are clear. Topical copper peptide products are available without a prescription as cosmetics. Injectable GHK-Cu is available with a prescription through licensed compounding pharmacies, fully restored to Category 1 status since February 2026. Research-grade peptides are purchasable for laboratory use.
The FDA approval framework was designed for patentable pharmaceutical products. Natural peptides like GHK-Cu, BPC-157, and TB-500 do not fit that model. Understanding this distinction lets you make an informed decision based on the actual regulatory landscape rather than a misleading binary of "approved" versus "unapproved."
For dosage protocols, see our GHK-Cu injection dosage guide. For reconstitution, use the peptide reconstitution calculator. For the full benefit profile, read GHK-Cu benefits. For the broader regulatory picture, see are peptides legal and FDA peptide crackdown 2026.
Related Articles
Is Tesamorelin FDA Approved? Status & Facts
Is tesamorelin FDA approved? Yes, as Egrifta for HIV lipodystrophy (2010). Approval timeline, off-label rules, and GH peptide comparison.
Does Compounded Tirzepatide Work? Real Data
Does compounded tirzepatide work? Review SURMOUNT data, salt form differences (base, acetate, sodium), potency gaps, and FDA status in 2026.
What Do Peptides Do for Skin? Benefits & Types
Peptides boost collagen by up to 65%, reduce wrinkles, and strengthen the skin barrier. Which types work, how to use them, and clinical evidence.
Does GHK-Cu Block DHT? Science Explained
Does GHK-Cu block DHT? Copper ions inhibit type 1 5-alpha reductase by up to 90% in vitro. Comparison to finasteride and practical limitations.