The Wolverine peptide stack combines BPC-157 (250-500 mcg daily) and TB-500 (2-5 mg twice weekly) for injury recovery. BPC-157 builds new blood vessels at the injury site through VEGF upregulation. TB-500 mobilizes repair cells systemically through actin regulation. The name comes from the X-Men character's regenerative healing. Neither peptide is FDA-approved. No study has tested the combination.
| Feature | BPC-157 | TB-500 | Combined Stack |
|---|---|---|---|
| Mechanism | Angiogenesis, NO system | Actin regulation, cell migration | Complementary pathways |
| Best for | Localized injury (one site) | Systemic repair (multiple sites) | Full-spectrum healing |
| Standard dose | 250-500 mcg/day | 2-5 mg 2x/week | Both at standard doses |
| Half-life | ~4 hours | ~2-7 days | N/A |
| Route | Subcutaneous (near injury) | Subcutaneous (any site) | Both subcutaneous |
| Monthly cost | $40-80 | $60-120 | $100-200 |
| Human trials | 3 pilot studies | Phase 2 (dry eye, wounds) | Zero combination studies |
The Wolverine stack is the most popular peptide pairing in the recovery community. The complementary mechanism theory is plausible. The evidence gap is real. This guide covers dosing protocols, the science behind each peptide, blend vs. separate purchasing, and an honest assessment of what the evidence supports.
For individual peptide details, see our BPC-157 benefits guide and what TB-500 does. This is educational content about research compounds.
Get your custom peptide protocol:
- Tailored to your body and goals
- Precise dosing and cycle length
- Safe stacking combinations
- Backed by peer-reviewed studies
- Ready in under 2 minutes
Why These Two Peptides Together
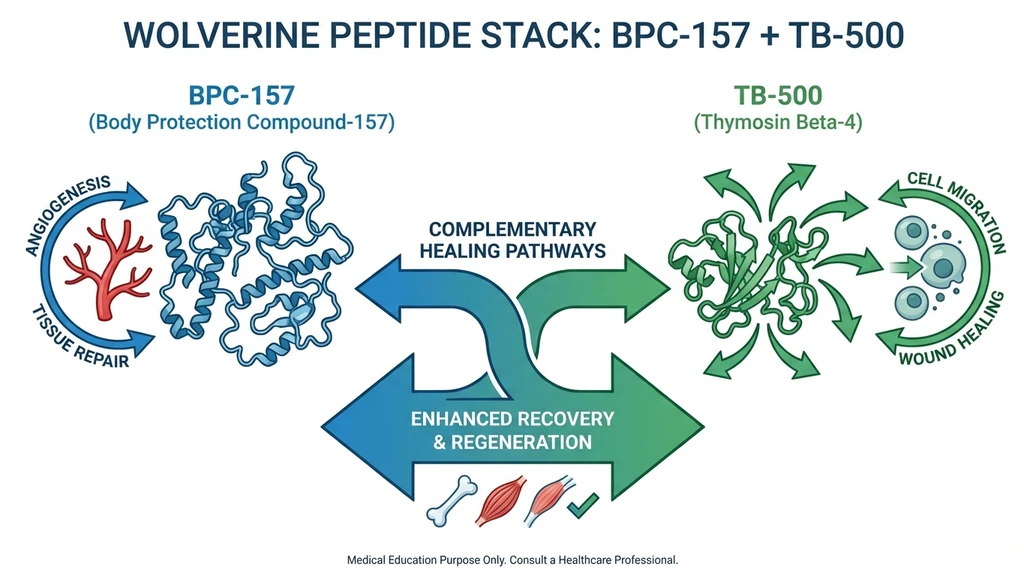
Joe Rogan called BPC-157 "Wolverine shit" on his podcast after it resolved his elbow tendinitis in roughly two weeks. The community later paired it with TB-500 to create the "stack." The Wolverine name stuck because of the X-Men character's regenerative healing ability. The combination spread through fitness forums, biohacking communities, and eventually clinical anti-aging practices.
BPC-157 and TB-500 target different phases of tissue repair through different biological pathways. This is why the community pairs them rather than doubling down on one compound.
BPC-157 builds the infrastructure. It upregulates vascular endothelial growth factor, creating new capillaries at the injury site. More blood supply means more oxygen, nutrients, and immune cells reaching damaged tissue. It works best when injected near the injury because its 4-hour half-life limits systemic distribution.
TB-500 sends the repair cells. It regulates actin polymerization, which controls how cells move and restructure. TB-500 promotes endothelial cell migration, reduces NF-kB-driven inflammation, and supports stem cell mobilization. Its 2-7 day half-life means it distributes systemically, reaching multiple injury sites regardless of injection location.
The complementary theory: BPC-157 builds the roads (blood vessels). TB-500 sends the repair trucks (migrating cells). One handles local infrastructure. The other handles systemic logistics. The pathways do not overlap, which is why researchers hypothesize synergy rather than redundancy.
This synergy has never been tested in a controlled study. Every claim about the combination is extrapolated from individual compound research. For a direct comparison, see our BPC-157 vs TB-500 guide.
How BPC-157 Works
BPC-157 is a 15-amino-acid peptide derived from a protective protein found in human gastric juice. It is stable in stomach acid, which distinguishes it from most peptides. Its primary repair mechanism centers on building new blood supply to damaged tissue.
VEGF upregulation and angiogenesis. BPC-157 stimulates vascular endothelial growth factor through the VEGFR2-Akt-eNOS signaling pathway (Hsieh et al., J Mol Med, 2017, PMID: 27847966). This triggers the formation of new capillaries at the injury site. More blood vessels mean more oxygen, nutrients, and immune cells reaching damaged tissue. In rat Achilles tendon transection models, BPC-157 treatment resulted in faster restoration of tendon continuity and improved biomechanical properties compared to controls.
Nitric oxide modulation. BPC-157 interacts with the nitric oxide system, influencing blood flow and inflammatory signaling at the injury site (Sikiric et al., Sci Rep, 2020, PMID: 33144807). This dual action on both VEGF and NO pathways may explain its broad efficacy across tissue types in animal models. BPC-157 appears to function as a stabilizer of the NO system rather than a simple agonist or antagonist, which may explain why it shows protective effects in both NO-excess and NO-depletion scenarios.
Fibroblast stimulation. BPC-157 promotes fibroblast proliferation, the cells responsible for producing collagen and rebuilding the structural matrix of connective tissue. This is particularly relevant for tendon and ligament injuries where collagen deposition is the rate-limiting step. In preclinical tendon-to-bone healing models, BPC-157 increased type I collagen expression at the repair site within the first two weeks.
Half-life and injection strategy. BPC-157 has an estimated half-life of approximately 4 hours. This short window limits systemic distribution. Local injection near the injury site is preferred to maximize concentration where repair is needed. For a full breakdown of BPC-157's mechanisms, see our BPC-157 benefits guide.
How TB-500 Works
TB-500 is a synthetic form of thymosin beta-4, a naturally occurring protein involved in cell migration and tissue repair. It works systemically rather than locally, making it the complement to BPC-157's site-specific action.
TB-500 vs. thymosin beta-4 clarification. TB-500 is technically a 43-amino-acid fragment of the full-length 44-amino-acid thymosin beta-4 protein. In practice, most vendors sell full-length thymosin beta-4 and label it "TB-500." The biological activity is similar. Ask the vendor for a certificate of analysis specifying the exact amino acid sequence.
Actin regulation and cell migration. TB-500 regulates actin polymerization, the process that controls how cells move, restructure, and divide. By sequestering G-actin monomers, TB-500 promotes cytoskeletal reorganization. This enables endothelial cells, keratinocytes, and other repair cells to migrate toward damaged tissue. The central actin-binding domain (amino acids 17-23) is the sequence responsible for this activity.
Anti-inflammatory action. TB-500 shifts macrophage polarization from the pro-inflammatory M1 phenotype toward the repair-oriented M2 phenotype. It modulates NF-kB signaling, reducing the inflammatory cascade that can delay healing when it persists beyond the acute phase. This is particularly relevant for chronic injuries where persistent low-grade inflammation creates a hostile environment for tissue repair. By shifting the immune response from destruction to reconstruction, TB-500 creates conditions favorable for new tissue formation.
Wound healing data. In a dermal wound model, topical thymosin beta-4 application increased reepithelialization by 42% at day 4 and 61% at day 7 compared to controls (Malinda et al., J Invest Dermatol, 1999, PMID: 10469335). This remains one of the most cited studies for TB-500's repair potential.
Half-life and injection strategy. TB-500 has an estimated half-life of 2 to 7 days. This long half-life means it distributes systemically regardless of injection location. Injection site does not matter for TB-500; most users inject subcutaneously in the abdomen for convenience. For more on TB-500's mechanisms, see our guide on what TB-500 does.
What the Evidence Actually Shows
Honest evidence grading for each component.
BPC-157: Strong preclinical, minimal human data. A 2025 systematic review screened 544 articles and included 36 studies: 35 preclinical and 1 clinical (Vasireddi et al., HSS Journal, PMID: 40756949). The single clinical study was a retrospective review of 12 patients with chronic knee pain; 7 of 12 reported pain relief lasting longer than 6 months. Earlier pilot studies include an interstitial cystitis trial (12 women, 2024) and an IV safety pilot (2 adults, 2025, PMID: 40131143). No Phase 3 trials exist. No registered trials are recruiting on ClinicalTrials.gov.
Single-lab concern. Over 80% of BPC-157 publications originate from Sikiric's group at the University of Zagreb. This concentration raises questions about independent replication. No other lab has reproduced the full range of effects reported by the Zagreb group. A few independent groups have confirmed specific mechanisms (e.g., VEGF upregulation, tendon healing in rats), but the sweeping multi-organ efficacy claims remain largely unreplicated. This does not invalidate the findings, but it is a significant limitation that readers should weigh when evaluating the evidence base.
TB-500: Moderate human evidence. Thymosin beta-4 (the parent molecule) has Phase 2 clinical trial data across multiple indications. A venous stasis ulcer trial (NCT00832091) and a pressure ulcer trial (NCT00598871) both showed accelerated healing by nearly a month compared to placebo. Safety profiles across both trials were comparable to placebo. A Phase 1 trial in healthy volunteers tested doses up to 25 mcg/kg with no serious adverse events (PMID: 34346165). Unlike BPC-157, thymosin beta-4 research comes from multiple independent labs and institutions worldwide, providing stronger confidence in the reproducibility of results.
The combination: Zero studies. No published study tests BPC-157 and TB-500 together in any model. The complementary mechanism theory is based on understanding each compound individually. The synergy is plausible but unproven. Until a controlled trial evaluates the combination, every claim about enhanced efficacy from pairing these peptides remains speculative.
For general peptide safety information, see our peptide safety guide and peptide stacking guide.
Dosing Protocols
Three protocol tiers based on experience level and injury severity.
Beginner Protocol (4-6 weeks)
| Peptide | Dose | Frequency | Injection Site |
|---|---|---|---|
| BPC-157 | 250 mcg | Once daily | Subcutaneous, near injury |
| TB-500 | 2 mg | Twice per week | Subcutaneous, abdomen |
Standard Protocol (6-8 weeks)
| Peptide | Dose | Frequency | Injection Site |
|---|---|---|---|
| BPC-157 | 500 mcg | Once daily | Near injury, rotate sides |
| TB-500 | 2.5 mg | 2x/week (loading 4 wks), then 1x/week | Abdomen |
Advanced Protocol (8-12 weeks)
| Peptide | Dose | Frequency | Injection Site |
|---|---|---|---|
| BPC-157 | 500 mcg | Twice daily (AM near injury, PM abdomen) | Split sites |
| TB-500 | 5 mg | 2x/week (loading 6 wks), then 2.5 mg 1x/week | Abdomen |
TB-500 protocols typically include a loading phase (higher frequency for 4-6 weeks) followed by a maintenance phase (reduced frequency). BPC-157 does not require loading because of its shorter half-life.
Taper protocol. After the standard 6-8 week protocol, reduce doses over 2-4 weeks rather than stopping abruptly. Reduce BPC-157 to half dose (125-250 mcg) for 1-2 weeks, then stop. Reduce TB-500 to once weekly for 2 weeks, then stop. Abrupt cessation is not harmful, but tapering may support continued tissue remodeling during the final repair phase.
Both peptides are reconstituted from lyophilized powder with bacteriostatic water. Use our reconstitution calculator for exact measurements. For injection technique, see our how to inject peptides guide.
Week-by-Week Timeline
What to expect during a standard 6-8 week Wolverine stack protocol. This timeline is extrapolated from preclinical data and community reports. No controlled human trial has measured these milestones for the combination.
| Week | What to Expect | Biological Process |
|---|---|---|
| 1-2 | Reduced sharp pain, warmth at injury site, less morning stiffness | Vascular reopening (BPC-157 VEGF), initial inflammation reduction (TB-500 NF-kB) |
| 3-4 | Improved range of motion, reduced swelling | Capillary maturation, fibroblast activation, collagen deposition begins |
| 5-6 | Functional improvement, return to light activity | Structural tissue remodeling, actin-driven cell migration completing |
| 7-8 | Near-baseline function for moderate injuries | Collagen consolidation, tensile strength approaching pre-injury levels |
| 9-12 | Continued remodeling (severe injuries only) | Full tissue maturation; taper off rather than abrupt stop |
Weeks 1-2 are primarily vascular and anti-inflammatory. BPC-157's VEGF upregulation reopens blood supply to the injury. TB-500's NF-kB modulation reduces chronic inflammation that may be blocking repair. The warmth some users report at the injection site likely reflects increased local blood flow.
Weeks 3-6 are the structural rebuilding phase. Fibroblasts lay down new collagen. Actin-driven cell migration fills in the damaged tissue matrix. Range of motion improvements typically become noticeable during this window.
Weeks 7-12 are consolidation. New collagen matures and cross-links, increasing tensile strength. Moderate injuries often reach near-baseline function by week 8. Severe injuries, particularly large tendon tears or post-surgical recovery, may require the full 12 weeks. Tapering during this phase supports the final remodeling without abrupt disruption.
Blend Products vs. Separate Vials
You can purchase BPC-157 and TB-500 as a pre-mixed blend or as separate vials. Each approach has trade-offs.
| Factor | Blend (Pre-Mixed) | Separate Vials |
|---|---|---|
| Convenience | High (one vial, one injection) | Lower (two reconstitutions, two injections) |
| Dose flexibility | Low (fixed ratio) | High (adjust each independently) |
| Local injection | Cannot split (both go to same site) | BPC near injury + TB-500 abdomen |
| Cost per week | $35-50/week | $25-40/week |
| Available formats | 10 mg (5+5), 20 mg (10+10) | Any size |
When to choose separate vials: If you have a specific injury and want BPC-157 injected near the site for maximum local effect. If you want to adjust doses independently based on response.
When to choose a blend: If you want convenience and are using the stack for general recovery or maintenance rather than targeting a specific injury.
The GLOW blend adds GHK-Cu as a third peptide for collagen cross-linking. If skin quality or scar reduction is also a goal, see our GLOW blend dosage guide and glow peptide dosage.
Injury-Specific Protocols
Different injuries respond to different protocol adjustments. The base dosing remains the same. The variables are injection site, duration, and whether BPC-157 benefits from local versus systemic delivery.
Tendon and Ligament Injuries
| Parameter | Recommendation | Rationale |
|---|---|---|
| BPC-157 dose | 500 mcg/day | Higher end for dense connective tissue |
| BPC-157 site | Subcutaneous, within 2 cm of tendon | Short half-life favors local delivery |
| TB-500 dose | 2.5 mg 2x/week (loading), 2.5 mg 1x/week (maintenance) | Systemic repair cell mobilization |
| Duration | 8-12 weeks | Tendon collagen turnover is slow (60-90 days) |
| Common targets | Achilles, patellar tendon, rotator cuff, tennis elbow, plantar fascia | All respond to angiogenesis + fibroblast stimulation |
Tendon injuries respond slower than muscle injuries. Tendons have poor blood supply (the core problem BPC-157 addresses through VEGF upregulation). Collagen fibers in tendons take 60 to 90 days to mature and cross-link. Cutting the protocol short at 4 weeks often results in incomplete repair and re-injury. The full 8-12 weeks allows collagen consolidation.
Muscle Injuries
| Parameter | Recommendation | Rationale |
|---|---|---|
| BPC-157 dose | 250-500 mcg/day | Standard range |
| BPC-157 site | Near the muscle belly, not the insertion | Muscles have better blood supply; less local targeting needed |
| TB-500 dose | 2 mg 2x/week | Standard loading |
| Duration | 4-6 weeks | Muscles heal faster than tendons |
| Common targets | Hamstring strain, quad tear, calf strain, bicep tear | Rapid response expected |
Muscle tissue is highly vascularized. BPC-157's angiogenic effect is still useful (restoring damaged capillaries), but the baseline blood supply is better than tendons. Most muscle strains show significant improvement within 3-4 weeks. Complete tears or post-surgical repair may require the full 8-week protocol.
Joint and Cartilage
| Parameter | Recommendation | Rationale |
|---|---|---|
| BPC-157 dose | 500 mcg/day | Higher dose for avascular cartilage |
| BPC-157 site | Subcutaneous near the joint capsule | As close to the cartilage as subcutaneous injection allows |
| TB-500 dose | 2.5-5 mg 2x/week | Higher end; systemic anti-inflammatory is key |
| Duration | 12+ weeks | Cartilage regeneration is extremely slow |
| Common targets | Knee osteoarthritis, hip degeneration, shoulder labrum | Manage expectations; peptides do not regrow cartilage |
Cartilage is avascular. It has no blood supply. This makes BPC-157's angiogenic mechanism less directly applicable to the cartilage itself, though improving peri-articular blood flow may support the joint environment. TB-500's anti-inflammatory action (NF-kB modulation) may be more relevant for joint protocols by reducing the chronic inflammation that accelerates cartilage degradation. Set realistic expectations. Peptides may reduce pain and slow degeneration. They do not regenerate lost cartilage in adults.
Post-Surgical Recovery
| Parameter | Recommendation | Rationale |
|---|---|---|
| BPC-157 dose | 500 mcg/day | Maximum standard dose |
| Start timing | 3-7 days post-surgery (consult surgeon) | Allow initial hemostasis before promoting angiogenesis |
| TB-500 dose | 5 mg 2x/week loading for 4-6 weeks | Maximum loading for systemic repair mobilization |
| Duration | 8-12 weeks | Aligns with standard surgical rehabilitation timelines |
| Contraindication | Do not start until surgeon clears it | VEGF upregulation could theoretically interfere with hemostasis |
Post-surgical use is the most common clinical application in anti-aging and sports medicine practices. The timing of initiation is critical. Starting too early (within 48 hours) risks interfering with the normal hemostatic response. Most practitioners recommend waiting until surgical drains are removed and initial wound closure is confirmed before starting the protocol.
For body-weight-based BPC-157 dosing, see our BPC-157 dosage for 200 lb male guide. For general injection technique, see our how to inject peptides guide.
Reconstitution Step-by-Step
Both BPC-157 and TB-500 ship as lyophilized (freeze-dried) powder that must be reconstituted with bacteriostatic water before injection.
BPC-157 Reconstitution (5 mg vial)
| Step | Action | Detail |
|---|---|---|
| 1 | Gather supplies | 5 mg BPC-157 vial, 2 mL bacteriostatic water, 1 mL insulin syringe, alcohol swabs |
| 2 | Add water | Draw 2 mL bacteriostatic water. Inject slowly along the vial wall. Do not spray directly onto the powder |
| 3 | Mix | Gently swirl the vial. Never shake peptides (denaturation risk) |
| 4 | Calculate | 2 mL water in 5 mg vial = 2.5 mg/mL = 2,500 mcg/mL. For 250 mcg dose: draw 10 units (0.10 mL). For 500 mcg dose: draw 20 units (0.20 mL) |
| 5 | Store | Refrigerate at 36-46F (2-8C). Use within 28 days |
TB-500 Reconstitution (5 mg vial)
| Step | Action | Detail |
|---|---|---|
| 1 | Gather supplies | 5 mg TB-500 vial, 1 mL bacteriostatic water, 1 mL insulin syringe, alcohol swabs |
| 2 | Add water | Draw 1 mL bacteriostatic water. Inject slowly along the vial wall |
| 3 | Mix | Gently swirl. Wait 2-3 minutes for full dissolution |
| 4 | Calculate | 1 mL water in 5 mg vial = 5 mg/mL. For 2 mg dose: draw 40 units (0.40 mL). For 2.5 mg dose: draw 50 units (0.50 mL). For 5 mg dose: use entire vial |
| 5 | Store | Refrigerate at 36-46F (2-8C). Use within 28 days |
Reconstitution tips:
- Add water slowly. Forceful injection causes foaming, which denatures the peptide.
- If powder sticks to the stopper, do not shake. Let the vial sit upright for 10 minutes. The powder will dissolve as the water wicks upward.
- Label each vial with the date of reconstitution. Discard after 28 days.
- Use a fresh needle for each injection. Reusing needles introduces bacteria into multi-use vials.
- Bacteriostatic water (not sterile water) is required for multi-use vials. The benzyl alcohol preservative prevents bacterial growth over the 28-day use period.
For exact calculations, use our reconstitution calculator. For general peptide storage guidance, see our how to store peptides guide and reconstituted peptide shelf life guide.
Stacking with Other Peptides
The Wolverine stack is sometimes expanded with additional peptides depending on the user's goals. These combinations are community-derived. No controlled studies exist for any multi-peptide stack.
| Add-On Peptide | Purpose | Typical Dose | Compatibility |
|---|---|---|---|
| GHK-Cu | Collagen cross-linking, skin healing | 1,500-2,000 mcg/day | High (this creates the GLOW blend) |
| CJC-1295/Ipamorelin | Growth hormone release for deep tissue repair | CJC 100 mcg + Ipa 100 mcg before bed | Moderate (GH supports repair but adds complexity) |
| Selank | Anxiety reduction during injury recovery | 250-500 mcg/day intranasal | Low interaction risk |
| GHK-Cu nasal spray | Non-injectable collagen support | Per product instructions | Low interaction risk |
| Pentadecapeptide BPC-157 oral | GI healing (concurrent gut issues) | 250-500 mcg oral, twice daily | Same compound, different route |
The Wolverine + GHK-Cu expansion (creating the GLOW blend) is the most common add-on. GHK-Cu promotes collagen types I and III cross-linking and modulates over 4,000 genes related to tissue regeneration (Pickart et al., PMC4508379). It complements BPC-157's vascular infrastructure and TB-500's cell mobilization with structural matrix support. See our glow peptide dosage guide for the three-peptide protocol.
The Wolverine + GH secretagogue stack adds CJC-1295/Ipamorelin for overnight growth hormone release. GH promotes IGF-1 production, which supports muscle protein synthesis and connective tissue repair. This combination is popular among athletes recovering from surgery who want to accelerate the rehabilitation timeline. The downside is increased cost ($150-250/month for the GH secretagogue) and additional injection burden.
When NOT to stack further:
- If this is your first peptide protocol, run BPC-157 and TB-500 alone first. Establish baseline response before adding variables.
- If you have any undiagnosed health conditions, additional peptides add unknown interaction risk.
- If cost is a concern, the core Wolverine stack ($100-200/month) delivers the primary healing benefit. Additional peptides offer diminishing marginal returns.
For a complete overview of peptide combinations, see our peptide stacking guide and peptide interaction checker.
2026 Regulatory Status
The regulatory landscape for BPC-157 and TB-500 shifted in early 2026 and continues to evolve.
BPC-157: Category 2 to Category 1 reclassification pending. In February 2026, HHS Secretary RFK Jr. announced that 14 of 19 peptides would move from FDA Category 2 (restricted from compounding) back to Category 1 (compoundable). BPC-157 is on the reclassification list. If finalized, this means licensed 503A compounding pharmacies could legally prepare BPC-157 with a physician's prescription. The formal FDA updated list had not been published as of March 2026 (source: Frier Levitt legal analysis).
TB-500: Category 2 status. TB-500 (thymosin beta-4) remains on the FDA Category 2 list. It was not included in the February 2026 reclassification announcement. This means it cannot be compounded by 503A pharmacies. Availability is limited to research chemical vendors and 503B outsourcing facilities operating under specific exemptions.
What this means for the Wolverine stack:
| Peptide | Current Status | Expected Change | Access Options |
|---|---|---|---|
| BPC-157 | Category 2 (pending reclassification) | Category 1 (compoundable with Rx) | Research vendors now; compounding pharmacy soon |
| TB-500 | Category 2 | No change announced | Research vendors only |
WADA prohibition. Both BPC-157 and TB-500 remain prohibited by the World Anti-Doping Agency at all times, in and out of competition. BPC-157 falls under S0 (non-approved substances). TB-500 falls under S2 (peptide hormones, growth factors). A positive test typically results in a 4-year suspension from competition. This prohibition applies regardless of whether the peptides become legally compoundable for medical use.
Practical implications. The reclassification of BPC-157 to Category 1 would legitimize one half of the Wolverine stack through the medical system. Patients could obtain pharmaceutical-grade BPC-157 with a prescription from any compounding pharmacy. TB-500 would still require sourcing from research peptide vendors without the quality assurance of a regulated pharmacy. This asymmetry may encourage some users to run BPC-157 alone through a prescription pathway.
For the full regulatory timeline, see our FDA peptide crackdown analysis, are peptides legal guide, and Peptide Sciences status.
Vendor Selection and Quality Control
Neither BPC-157 nor TB-500 has USP manufacturing standards. Product quality varies significantly between vendors. These verification steps reduce risk.
Certificate of Analysis (COA) checklist:
| COA Element | What to Look For | Red Flag |
|---|---|---|
| Amino acid sequence | Match to published sequence | Missing or "proprietary" sequence |
| Purity (HPLC) | 98%+ | Below 95% or not stated |
| Endotoxin (LAL) | < 5 EU/mg | Not tested or elevated |
| Mass spectrometry | Molecular weight matches target | Missing or inconsistent |
| Testing lab | Third-party (not in-house) | In-house only, no lab name |
| Batch number | Matches the vial you received | Generic COA not tied to specific batch |
Third-party testing. The most reliable vendors use independent analytical labs (e.g., Janoshik, Vanta Bio, or university-affiliated labs) for COA testing. In-house testing alone is insufficient because it cannot be independently verified. Request the COA before purchasing. Any vendor that refuses to provide a batch-specific COA should be avoided.
Vendor reliability signals:
- Consistent product availability (frequent stockouts may indicate unstable supply chain)
- Transparent origin information (country of synthesis, GMP status)
- Customer reviews across multiple platforms (not just their own website)
- Responsive customer support with peptide-specific knowledge
- Proper cold-chain shipping (insulated packaging, ice packs for temperature-sensitive peptides)
What to avoid:
- Vendors that make therapeutic claims or promise specific medical outcomes
- Products sold as "for human use" without FDA approval (legal risk)
- Unusually low prices (below $25 for a 5 mg vial of either peptide suggests quality compromise)
- Pre-mixed solutions (reconstituted peptides degrade faster; buy lyophilized powder and reconstitute yourself)
For vendor reviews, see our Polaris Peptides review and Transcend Peptides review. For general purchasing guidance, see our where to buy peptides 2026 guide.
Side Effects and Safety
Both peptides have favorable safety profiles based on available data.
| Side Effect | BPC-157 | TB-500 |
|---|---|---|
| Injection site redness | Common, mild | Common, mild |
| Headache | Occasional | 10-15% |
| Nausea | Rare | 5-10% |
| Fatigue/lethargy | Rare | 10-15% |
| Old injury flare | Reported anecdotally | Reported anecdotally |
Cancer pre-screening. Both peptides are pro-angiogenic. BPC-157 upregulates VEGF. TB-500 promotes cell migration and proliferation. This raises a theoretical concern for individuals with undiagnosed malignancies, as new blood vessel formation could support tumor growth. Some clinics require oncological screening before starting the protocol. This risk is theoretical and has not been confirmed in human studies, but it is the primary medical contraindication.
NSAID interaction. No clinical data exists on this interaction. NSAIDs suppress prostaglandin synthesis, which overlaps with BPC-157's inflammatory signaling pathways. Some practitioners advise avoiding NSAIDs during the protocol to prevent blunting the healing response. Acetaminophen is generally considered a safer pain management alternative during the stack.
Product purity risks. Neither BPC-157 nor TB-500 has USP manufacturing standards. The peptide supply chain involves unregulated manufacturing with significant batch variability. Third-party certificate of analysis (COA) testing is the only quality control available. Request COAs specifying amino acid sequence, purity percentage, and endotoxin levels before purchasing from any vendor.
Regulatory status: Neither peptide is FDA-approved. BPC-157 is classified as FDA Category 2 (cannot be compounded by licensed pharmacies). Both are prohibited by WADA at all times. A positive test carries a 4-year ban.
For more on individual side effects, see our TB-500 side effects guide and BPC-157 liver safety. For legal context, see are peptides legal and FDA peptide crackdown.
Frequently Asked Questions
What is the Wolverine peptide stack?
The combination of BPC-157 and TB-500 peptides used together for injury recovery. BPC-157 builds blood vessels at the injury site while TB-500 mobilizes repair cells systemically. The name references Wolverine's superhuman healing ability from X-Men. Neither peptide is FDA-approved.
How long does the Wolverine stack take to work?
Most users report initial improvements in 1-2 weeks, with significant progress by 4-6 weeks. Animal studies show marker changes within days. Full tissue remodeling may take 8-12 weeks. Standard protocols run 6-8 weeks.
Is there scientific proof the Wolverine stack works?
Each peptide individually has supporting evidence. BPC-157 has 100+ animal studies and 3 small human pilots. TB-500 has Phase 2 human trials for dry eye and wounds. No study has tested the two in combination. The synergy is theoretically plausible but unproven.
Should I buy a blend or separate vials?
Separate vials for targeted injury recovery (BPC-157 near the injury, TB-500 in abdomen). Blends for convenience and general recovery. Separate vials are typically cheaper and allow independent dose adjustment. Blends require one reconstitution and one injection.
Is the Wolverine stack banned in sports?
Yes. Both BPC-157 and TB-500 are prohibited by WADA at all times, in and out of competition. BPC-157 falls under unapproved substances (S0). TB-500 falls under growth factors. A positive test typically results in a 4-year ban.
How much does the Wolverine stack cost per month?
Approximately $100-200 per month for research-grade peptides bought separately. Pre-mixed blends cost slightly more. Clinic-administered protocols run $300-600+ per month. Separate vials offer the best value with full dose control.
What is the difference between the Wolverine stack and the GLOW blend?
The Wolverine stack is BPC-157 + TB-500 (two peptides). The GLOW blend adds GHK-Cu as a third peptide for collagen cross-linking and skin rejuvenation. The Wolverine stack focuses on tissue repair. The GLOW blend adds a cosmetic/skin quality component.
Can I take BPC-157 and TB-500 at the same time?
Yes. Many users inject both during the same session. BPC-157 is typically injected subcutaneously near the injury site. TB-500 is injected in the abdomen. They target different biological pathways with no known interaction.
Can I take NSAIDs while on the Wolverine stack?
No clinical data exists on this interaction. NSAIDs suppress prostaglandin synthesis, which overlaps with BPC-157's inflammatory signaling. Some practitioners advise avoiding NSAIDs during the protocol to prevent blunting the healing response. Acetaminophen is generally considered safer for pain management during the stack.
What is the difference between TB-500 and thymosin beta-4?
TB-500 is a 43-amino-acid fragment of the full 44-amino-acid thymosin beta-4 protein. Most vendors sell full-length thymosin beta-4 labeled as TB-500. Biological activity is similar. Ask your vendor for a certificate of analysis specifying the exact amino acid sequence.
The Bottom Line
The Wolverine stack pairs BPC-157's local angiogenesis with TB-500's systemic cell mobilization. BPC-157 builds blood vessels at the injury site. TB-500 sends repair cells through the body. The complementary mechanism theory is sound. Zero combination studies exist to confirm the synergy.
Each peptide individually has data. BPC-157 has 100+ animal studies and 3 small human pilots, though 80%+ of publications originate from a single lab group in Zagreb. TB-500 has Phase 2 human trial data showing accelerated wound healing (42-61% faster reepithelialization) and a safety profile comparable to placebo. The combination is the most popular peptide stack in the recovery community, but popularity is not proof.
The practical reality: tendon and ligament injuries need 8-12 weeks. Muscle injuries respond in 4-6 weeks. Post-surgical recovery should start 3-7 days after surgeon clearance. Cartilage injuries require managed expectations. Separate vials give you dose flexibility and local BPC-157 injection. Blends offer convenience.
The 2026 reclassification of BPC-157 to Category 1 (pending FDA publication) may bring pharmaceutical-grade BPC-157 into the regulated compounding pharmacy system. TB-500 remains Category 2. Both remain prohibited by WADA.
For individual peptide dosing, see our BPC-157 dosage guide and TB-500 dosage guide. Use our reconstitution calculator for preparation and our cost calculator for treatment planning. For the three-peptide version, see our glow peptide dosage guide.
This is educational content about research compounds. Neither BPC-157 nor TB-500 is FDA-approved. Consult a healthcare provider before using any peptide.
Helpful Tools
Related Articles
Best Peptide Stacks: Charts & Protocols
Science-backed peptide stacking guide with compatibility charts, dosage protocols, and best stacks for healing, muscle growth, fat loss, and anti-aging.
BPC-157 Dosage Per Body Weight (Chart)
BPC-157 dosage by body weight: 1.6-10 mcg/kg. Full chart for 120-280 lbs, injection vs oral doses, and why most use flat 250-500 mcg.
Hims Weight Loss Reviews 2026: Pricing & Verdict
Hims weight loss reviews 2026: GLP-1s from $149/mo plus $149 membership. Branded Wegovy and Ozempic, Trustpilot 3.5/5. Pricing and red flags.
Found Weight Loss Reviews 2026: Pricing & Verdict
Found weight loss reviews 2026: GLP-1s from $129/mo self-pay or $49/mo with insurance. Trustpilot 3.6/5. Pricing, meds, and honest verdict.